Credit: (Photo: Jacobs University/Khaleel Assaf)
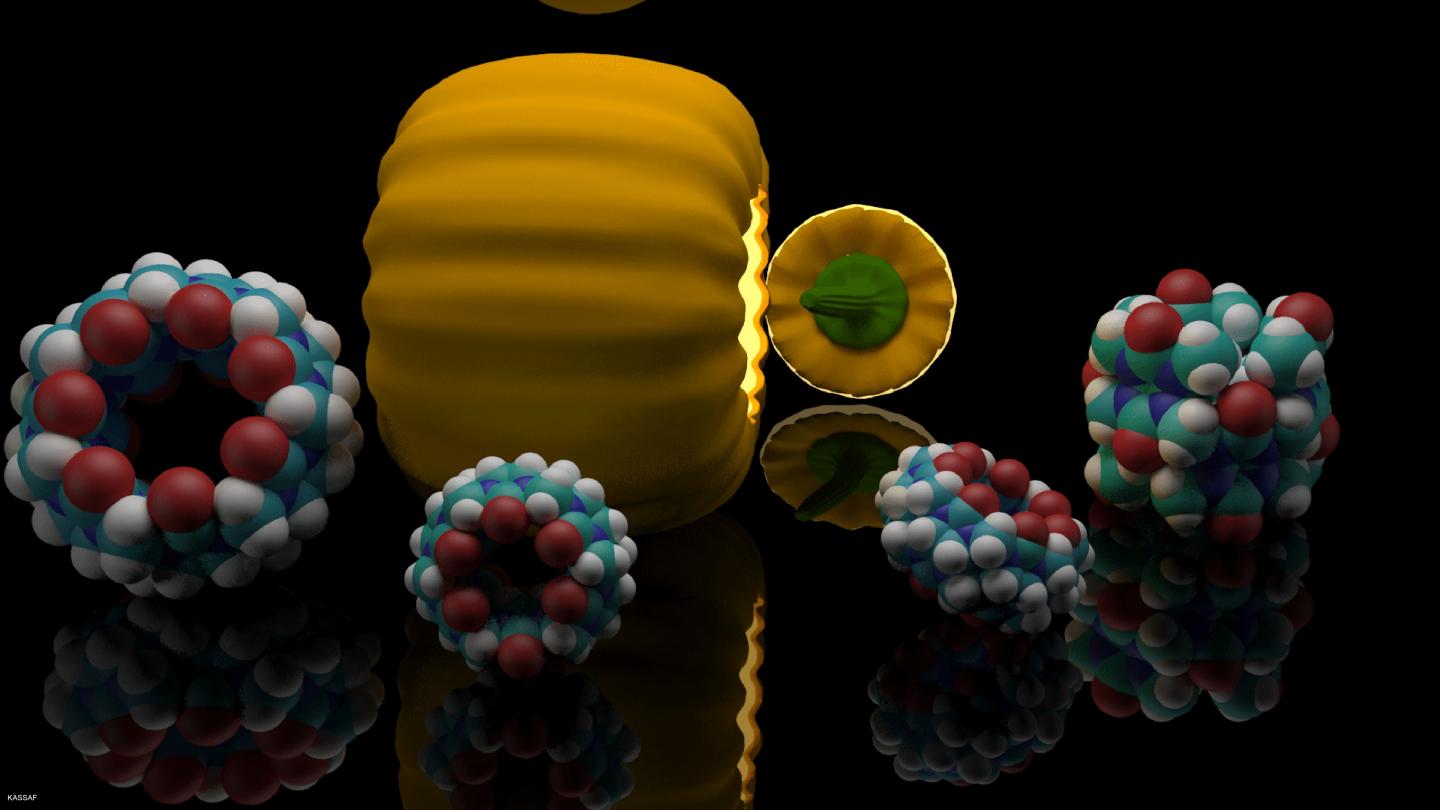
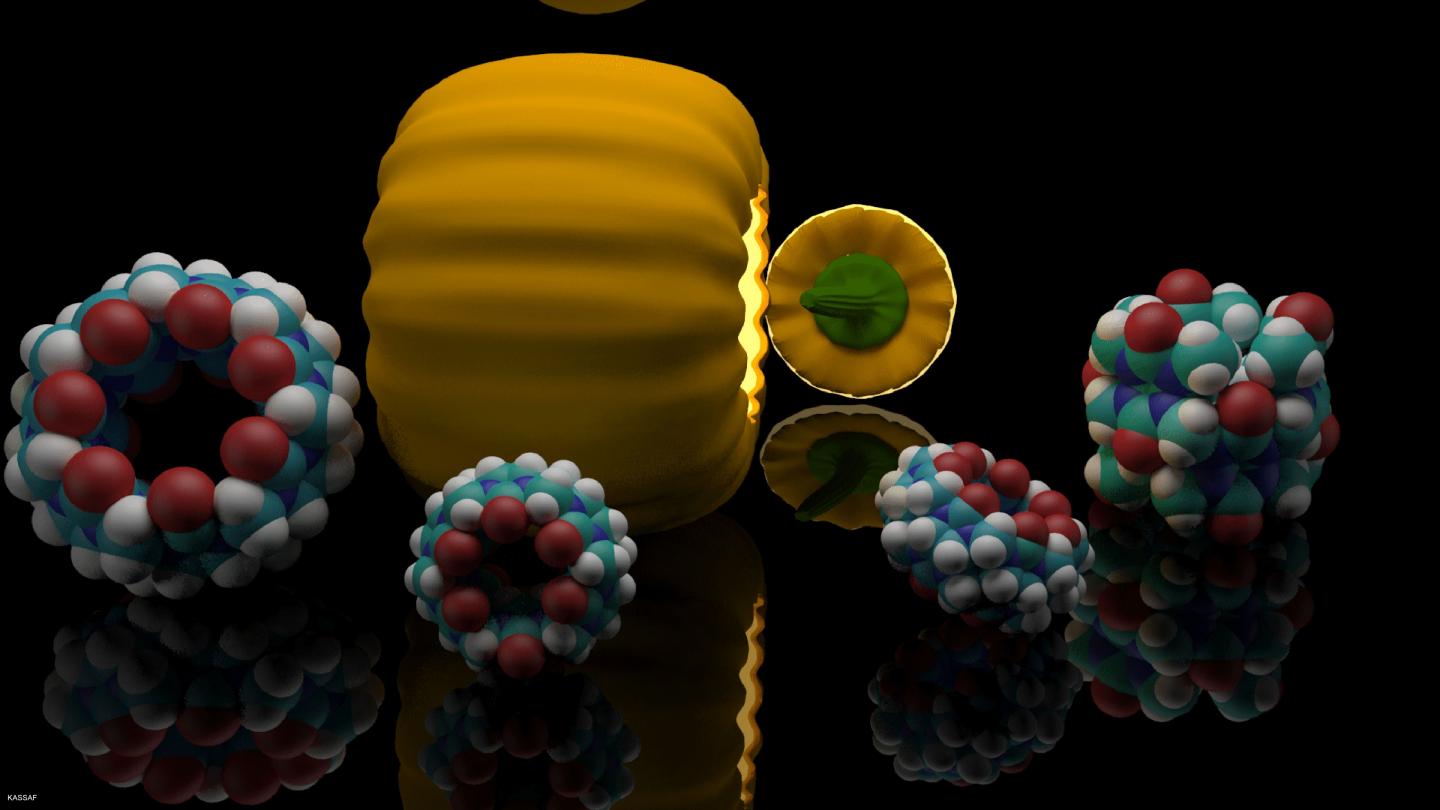
Synthetic hosts are to transport medical substances and hormones into the body and to release them at specific points. They enclose the active substances in a cavity. In case of steroids, this task has been accomplished mainly by ring-shaped glucose molecules so far. Now, scientists of KIT and Jacobs University Bremen have discovered a new class of host molecules, namely, barrel-shaped cucurbiturils. They can make hardly soluble steroids, such as cortisone or estradiol, act more gently and more efficiently. DOI: 10.1021/jacs.6b07655
"We found that the host class of cucurbiturils has a higher affinity to steroids used for medical purposes than cyclodextrines," Frank Biedermann, scientist of the KIT Institute of Nanotechnology, explains. The ring-shaped glucose of cyclodextrine is a relatively large molecule that can be adapted easily thanks to its flexible shape. On the other hand, it also collapses more easily. To produce the required water solubility, an increased dose of the active substance and transporter medium is required. This increases undesired side effects of the respective medicine. In addition, cyclodextrines preferably bind to thinner molecule chains, such as cholesterols that are irrelevant as active substances.
Based on experiments with the hormones testosterone and estradiol, the inflammation inhibitor cortisol, and the muscle relaxants pancuronium and vercuronium, the experts proved that steroid cucurbiturils are far more stable and more strongly increase water solubility of their host molecule. In addition, they can act as deposit of the active substance, because they also remain stable in blood serum and gastric acid and release steroids more slowly in the body. The new host group is biocompatible and can be applied at a reduced dose and more selectively. As a consequence, medicine based on steroids can act much better, side effects can be reduced, and production costs decrease.
"With the help of cucurbiturils, new and more efficient formulations of steroid substances might be developed in the future," Werner Nau, expert for supramolecular chemistry at Jacobs University Bremen, says. But not only pharmacology, also biological fundamental research will profit from the new substance transporters in the opinion of both scientists. In combination with an indicator dye, cucurbiturils allow for observing in real time the interaction between steroids and enzymes on their way through the body.
A new research project just started by Biedermann at KIT is aimed at showing the variety of ways in which these molecules can be used. The scientist wants to demonstrate that cucurbiturils do not only mobilize steroids, but can also immobilize them again e.g. when they enter the groundwater with body excretions.
###
Publication of the results: J.Am.Chem.Soc. 2016, 138, 13022-13029; http://pubs.acs.org/doi/abs/10.1021/jacs.6b07655
Kosta Schinarakis, KIT, Science Scout, Phone: +49 721 608 41956, Fax: +49 721 608 43658, Email: [email protected]
Karlsruhe Institute of Technology (KIT) pools its three core tasks of research, higher education, and innovation in a mission. With about 9,300 employees and 25,000 students, KIT is one of the big institutions of research and higher education in natural sciences and engineering in Europe.
KIT – The Research University in the Helmholtz Association
Since 2010, the KIT has been certified as a family-friendly university.
Media Contact
Monika Landgraf
[email protected]
49-721-608-47414
@KITKarlsruhe
http://www.kit.edu/index.php
############
Story Source: Materials provided by Scienmag