Credit: Yun Li and Julien Muffat
CAMBRIDGE, Mass. (December 29, 2016) – One of the most significant ways in which the human brain is unique is the size and structure of the cerebral cortex. But what drives the growth of the human cortex, likely the foundation for our unique intellectual abilities?
In research published in the journal Cell Stem Cell–in a study entitled, Induction of expansion and folding in human cerebral organoids–researchers at Whitehead Institute provide insight into a specific gene pathway that appears to regulate the growth, structure, and organization of the human cortex. They also demonstrate that 3D human cerebral organoids–miniature, lab-grown versions of specific brain structures–can be effective in modeling the molecular, cellular, and anatomical processes of human brain development. And they suggest a new path for identifying the cells affected by Zika virus.
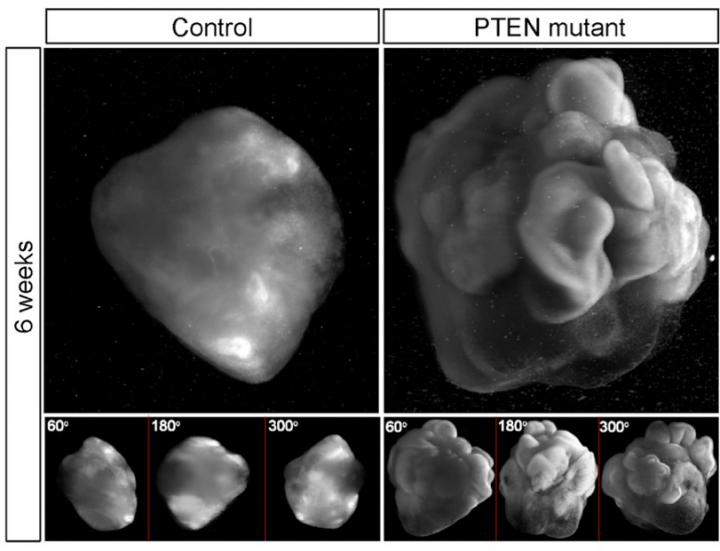
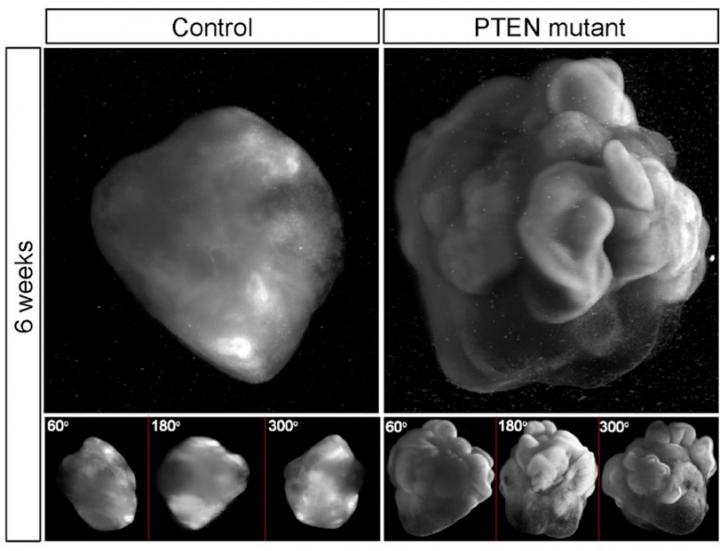
"We found that increased proliferation of neural progenitor cells (NPs) induces expansion of cortical tissue and cortical folding in human cerebral organoids," says Yun Li, a lead author of study and post-doctoral researcher at Whitehead Institute. "Further, we determined that deleting the PTEN gene allows increased growth factor signaling in the cell, unleashing its growth potential, and stimulating proliferation."
These findings lend support to the notion that an increase in the proliferative potential of NPs contributes to the expansion of the human cerebral neocortex, and the emergence of surface folding.
With normal NPs, the human organoid developed into relatively small cell clusters with smooth surface appearance, displaying some features of very early development of a human cortex. However, deleting PTEN allowed the progenitor population to continue expanding and delayed their differentiation into specific kinds of neurons–both key features of the developing human cortex. "Because the PTEN mutant NPs experienced more rounds of division and retained their progenitor state for an extended period, the organoids grew significantly larger and had substantially folded cortical tissue," explains Julien Muffat, also a lead author and post-doctoral researcher at Whitehead Institute.
In contrast, they found that while PTEN deletion in mouse cells does create a somewhat larger than normal organoid, it does not lead to significant NP expansion or to folding. "Previous studies have suggested that abnormal variation in PTEN expression may play an important role in driving brain development conditions leading to syndromes such as Autism Spectrum Disorders," says Rudolf Jaenisch, Founding Member of Whitehead Institute and senior author of the study. "Our findings suggest that the PTEN pathway is also an important mechanism for controlling brain-structure differences observed between species."
The Whitehead investigators chose to focus on the PTEN gene because it had previously been shown to have some function in cortical development and to have a role in regulating progenitor cells of various lineages. Notably PTEN loss-of-function mutations have been associated with human macrocephaly.
In this study, deletion of the PTEN gene increased activation of the PI3K-AKT pathway and thereby enhanced AKT activity in the human NPs comprising the 3D human cerebral organoids; it promoted cell cycle re-entry and transiently delayed neuronal differentiation, resulting in a marked expansion of the radial glia and intermediate progenitor population. Validating the molecular mechanism at work with PTEN, the investigators used pharmacological AKT inhibitors to reverse the effect of the PTEN deletion. They also found that they could regulate the degree of expansion and folding by tuning the strength of AKT signaling–with reduced signaling resulting in smaller and smooth organoids, and increased signaling producing larger and more folded organoids.
Finally, the researchers utilized the 3D human cerebral organoid system to show that infection with Zika virus impairs cortical growth and folding. In the organoids, Zika infection at the onset of surface folding (day 19 of development) led to widespread apoptosis; and, ten days later, it had severely hampered organoid growth and surface folding. Zika infection of 4-week-old organoids, showed that PTEN mutant organoids were much more susceptible to infection than normal control organoids; notably, they showed increased apoptosis and decreased proliferation of progenitor cells.
"Although not an original goal of our study, we have demonstrated that 3D human cortical organoids can be very effective for Zika modeling–better enabling researchers to observe how human brain tissue reacts to the infection and to test potential treatments," Li says.
###
This work was supported by a Simons Postdoctoral Fellowship, an International Rett Syndrome Foundation Postdoctoral Fellowship, and a NARSAD Young Investigator Grant from the Brain & Behavior Research Foundation (YL); support from the European Leukodystrophy Association and a NARSAD Young Investigator Grant from the Brain & Behavior Research Foundation (JM); a Jerome and Florence Brill Graduate Student Fellowship (AO); NIH grants HD 045022, R01-MH104610, R01-NS088538, and grants from the Simons Foundation (SFARI 204106 and SFLIFE 286977), the ELA foundation, the Emerald Foundation, Biogen, and the Simons Center for the Social Brain at MIT (R.J. laboratory); and NIH grant AI100190 (L.G. laboratory).
Rudolf Jaenisch's primary affiliation is with Whitehead Institute for Biomedical Research, where his laboratory is located and all his research is conducted. He is also a professor of biology at Massachusetts Institute of Technology.
Yun Li is a post-doctoral researcher in the Jaenisch lab at Whitehead Institute.
Julien Muffat is a post-doctoral researcher in the Jaenisch lab at Whitehead Institute.
Full Citation:
"Induction of expansion and folding in human cerebral organoids"
Cell: Stem Cell, December 29, 2016
Yun Li 1,8, Julien Muffat 1,8, Attya Omer 1, Irene Bosch 2, Madeline A. Lancaster 3, Mriganka Sur 4, Lee Gehrke 2,5,6, Juergen A. Knoblich 3, and Rudolf Jaenisch 1,7
1 The Whitehead Institute for Biomedical Research, 9 Cambridge Center, Cambridge, MA 02142, USA
2 Institute for Medical Engineering and Science, Massachusetts Institute of Technology, Cambridge MA 02139, USA
3 Institute of Molecular Biotechnology of the Austrian Academy of Sciences, 1030 Vienna, Austria
4 The Picower Institute for learning and memory, Cambridge MA 02139, USA
5 Department of Microbiology and Immunobiology, Harvard Medical School, Boston 02115, USA
6 Harvard-MIT Program in Health Sciences and Technology, Cambridge, MA 02139, USA
7 Department of Biology, Massachusetts Institute of Technology, 31 Ames Street, Cambridge, MA 02139, USA
8 These authors contributed equally
Whitehead Institute is a world-renowned non-profit research institution dedicated to improving human health through basic biomedical research. Wholly independent in its governance, finances, and research programs, Whitehead shares a close affiliation with Massachusetts Institute of Technology through its faculty, who hold joint MIT appointments.
Media Contact
Merrill Meadow
[email protected]
339-223-7307
@WhiteheadInst
http://www.wi.mit.edu/index.html
############
Story Source: Materials provided by Scienmag