Scientists from Queen Mary University of London have made a breakthrough in developing a new therapy for advanced bladder cancer – for which there have been no major treatment advances in the past 30 years.
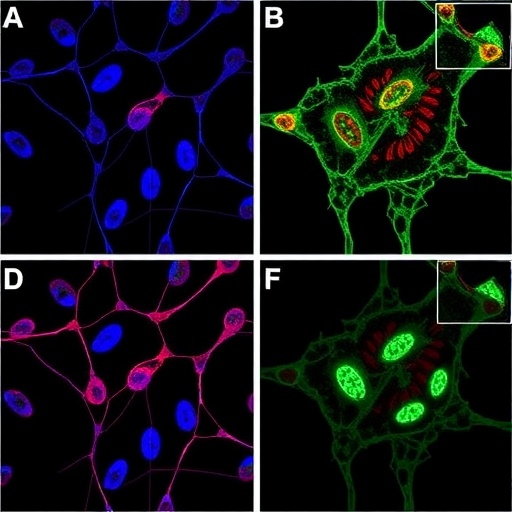
Published yesterday in Nature, the study examined an antibody (MPDL3280A) which blocks a protein (PD-L1) thought to help cancer cells evade immune detection.
In a phase one, multi-centre international clinical trial, 68 patients with advanced bladder cancer (who had failed all other standard treatments such as chemotherapy) received MPDL3280A, a cancer immunotherapy medicine being developed by Roche. In addition, patients were all tested for the protein PD-L1 and around 30 were identified as having PD-L1 positive tumours.
After six weeks of treatment, 43 per cent of PD-L1-positive patients found their tumour had shrunk. This rose to 52 per cent after 12 weeks of follow up. In two of these patients (7 per cent) radiological imaging found no evidence of the cancer at all following the treatment. Among PD-L1 negative patients, 11 percent responded positively to treatment too.
Patients who had a positive response to treatment found the benefits were prolonged, and safety results were also encouraging, with fatigue and loss of appetite most commonly reported as side effects.
The early results of this trial are so promising, the MPDL3280A antibody drug has been given breakthrough therapy designation status by the U.S. FDA.
Dr Tom Powles, Lead Author and Consultant Medical Oncologist, Barts Cancer Institute, Queen Mary University of London, comments: “This study is a hugely exciting step forward in the search for alternative advanced bladder cancer treatment. For decades chemotherapy has been the only option, with a poor outcome and many patients too ill to cope with it. Not only has this investigational drug had a striking response rate, we can target this therapy for patients by screening specific protein PD-L1.
“We now need larger trials to confirm our findings, and as this drug has been given breakthrough designation status by the FDA, we hope to fast track this process so we can begin to give hope to the thousands of people affected by advanced bladder cancer each year.”
Bladder cancer is the 7th most common cancer in the UK and around 10% of diagnoses are advanced (meaning the cancer has already spread to another part of the body). This makes it very difficult to treat, with chemotherapy the only option. On average patients live for 12 – 18 months following diagnosis, with many choosing to forgo chemotherapy due to its toxicity and limited survival benefit.
Story Source:
The above story is based on materials provided by Queen Mary, University of London.