Credit: Courtesy of Kenji Sugioka
EUGENE, Ore. — Jan. 30, 2017 — Researchers from three U.S. universities have identified, using roundworms, the earliest-acting protein known to duplicate the centriole, a tiny cylinder-shaped structure that is a key component of the machinery that organizes cell division in animals.
When the protein is damaged, cell division goes awry in ways that mirror conditions associated with human cancers, microcephaly and ciliopathies such as Bardet-Biedl and oral-facial-digital syndromes that involve myriad abnormalities throughout the body.
The discovery of the protein in C-elegans, a tiny nematode, is detailed in a paper published in January by the journal eLife. The mutant protein, dubbed sas-7, was found 17 years ago in the lab of co-author Bruce Bowerman, who now heads of the Department of Biology at the University of Oregon.
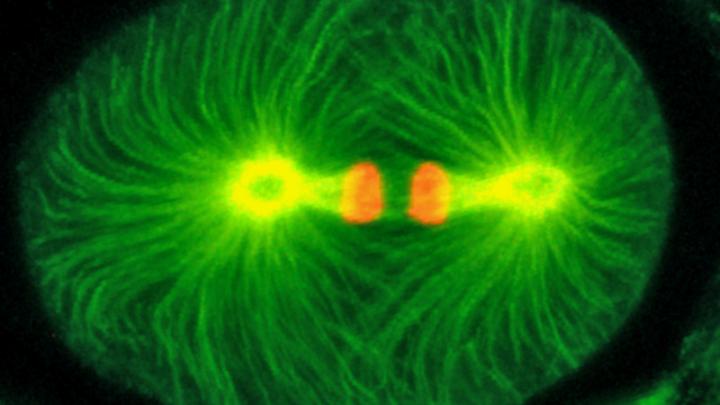
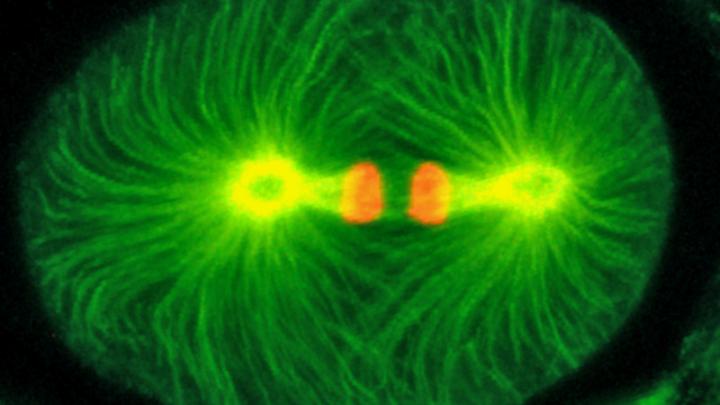
Using high-resolution transmission electron microscopy in collaboration with James Priess at the Fred Hutchinson Cancer Research Center in Seattle, the researchers were able to circle back to study centrioles at an ultrastructural level. The analysis identified a centriolar structure dependent on the protein — called a paddlewheel for its shape — that is vital for proper cell division in the worms. Similar structures have been observed in other animals but their functional importance was not known.
"The bottom line is that we've discovered a protein that acts at the earliest step known in centriole duplication," said Bowerman, a member and former head of the UO Institute of Molecular Biology. "Finding this protein provides a key advance in our understanding centriole duplication, which is critical to cell division and cilia function in animals. We also have attained new structural insights into the molecular organization of these fascinating structures."
The research focused on mitosis, the process by which a single cell divides to make two daughter cells. Each daughter cell inherits identical copies of the organism's genetic code — its biological instruction manual.
Vital to cell division is the presence of two pairs of centrioles, located near the nucleus of each cell, that organize rigid fibers called microtubules into a bipolar structure. This bipolar structure, called the mitotic spindle, separates duplicated chromosomes such that each daughter receives one copy of each chromosome.
Just as the DNA in chromosomes must be duplicated before cell division, so are the centrioles, which are complex assemblies of many different proteins. While chromosome duplication is well understood, centriole duplication remains mysterious.
When sas-7 is damaged, centriole duplication fails and a cell has only one centriole. As a result, a monopolar spindle forms and chromosomes are not separated into daughter cells. In addition, the structural integrity of the centriole's paddlewheel is compromised. "We think this paddlewheel is an important structural component of centrioles and may be a key part of how centrioles duplicate," Bowerman said.
Co-lead author Danielle R. Hamill, now a professor of zoology at Ohio Wesleyan University, had found the mutant protein while she was a postdoctoral researcher at the UO. Over the years, she returned to the UO to work with Bowerman to keep probing the role of sas-7.
"There have been previously identified proteins, including some with similar names, sas-1 through 6, all of which were identified more than 20 years ago. All have since been found to be conserved in humans," Bowerman said.
Previously, one of those proteins discovered earlier, spd-2, had been identified as the earliest-acting protein in mitotic cell division. Bowerman's team, co-led by current UO postdoctoral researcher Kenji Sugioka, determined that sas-7 is active upstream from spd-2.
"C-elegans has been the one model organism where the pathway of centriole duplication has been worked out and found to be conserved in all animals," Bowerman said. "We don't know how sas-7 is doing its job, but it does provide us with a new player in this pathway and may help us understand how cells know how to divide into two over and over, and accurately."
###
Co-authors on the paper with Bowerman, Hamill, Priess and Sugioka were Joshua B. Lowery, a former doctoral student in the UO's Institute of Molecular Biology, and Marie E. McNeely, Molly Enrick, Alyssa C. Richter and Lauren E. Kiebler, former and current students of Hamill's at Ohio Wesleyan.
Separate National Institutes of Health grants to Hamill, Bowerman and Priess primarily funded the work. Sugioka was supported by a Human Frontier Science Program grant from the France-based International Human Frontier Science Program Organization and a Journal of Cell Science travel fellowship.
Source: Bruce Bowerman, professor and head, Department of Biology, 541-346-0853, [email protected], and Danielle Hamill, Department of Zoology, Ohio Wesleyan University, 740-368-3888, [email protected]
Note: The UO is equipped with an on-campus television studio with a point-of-origin Vyvx connection, which provides broadcast-quality video to networks worldwide via fiber optic network. There also is video access to satellite uplink and audio access to an ISDN codec for broadcast-quality radio interviews.
Links:
About Bowerman: http://molbio.uoregon.edu/bowerman/
Institute of Molecular Biology: http://molbio.uoregon.edu/
Department of Biology: http://biology.uoregon.edu/
Bowerman lab: http://www.molbio.uoregon.edu/~bowermanlab/
Media Contact
Jim Barlow
[email protected]
541-346-3481
@UOregonNews
http://around.uoregon.edu
############
Story Source: Materials provided by Scienmag