In a groundbreaking development that could redefine the therapeutic landscape for glioblastoma, researchers at the University of Virginia Comprehensive Cancer Center have identified a small molecule inhibitor targeting the gene responsible for this aggressive brain cancer. Glioblastoma multiforme (GBM), known for its rapid progression and dismal prognosis, has long resisted effective treatment, with median survival times stubbornly remaining around 15 months despite surgical, chemotherapeutic, and radiotherapeutic interventions. This novel approach, spearheaded by Dr. Hui Li and colleagues, could signal the advent of an entirely new class of targeted therapies for GBM.
Glioblastoma is notorious for its invasive nature, infiltrating surrounding brain tissue in a manner that complicates surgical excision and enables rapid recurrence. The heterogeneity and resilience of GBM tumors have rendered conventional treatment strategies largely palliative, extending life only modestly while often severely compromising patients’ quality of life. The lack of progress in treatment is partly attributed to the absence of druggable molecular targets unique to glioblastoma cells, underscoring the urgent need for innovative therapeutic modalities.
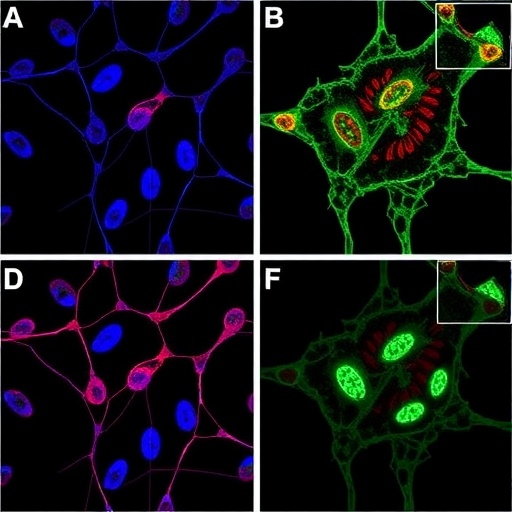
Dr. Hui Li’s team focused on an oncogene termed AVIL, which regulates cytoskeletal dynamics and cell morphology under physiological conditions. Their prior research in 2020 identified AVIL as a pivotal driver of glioblastoma oncogenesis, with its aberrant overexpression fostering malignant transformation and tumor proliferation. Importantly, AVIL activity was found to be markedly elevated in glioblastoma cells while being virtually undetectable in the normal brain, thereby representing a promising molecular vulnerability.
The current study deployed a high-throughput screening approach to sift through an extensive chemical library in search of small molecules capable of selectively inhibiting AVIL function. This methodology enabled the rapid evaluation of numerous compounds on glioblastoma cell cultures and mouse models. The resultant molecule demonstrated potent blockade of AVIL activity, impairing tumor growth and viability without damaging healthy brain tissue—a critical characteristic for any central nervous system-directed therapy.
Animal studies revealed that this molecule could cross the blood-brain barrier, a formidable obstacle in neuro-oncology drug development. The blood-brain barrier’s selective permeability often impedes drugs from reaching therapeutic concentrations within the brain parenchyma, severely limiting treatment options for brain malignancies. The ability of the AVIL inhibitor to penetrate this barrier and accumulate in the CNS substantiates its potential as a viable oral therapeutic.
Equally notable is the molecule’s safety profile observed in vivo. Unlike traditional chemotherapy and radiation, which induce widespread cytotoxicity, the AVIL inhibitor’s specificity for glioblastoma cells minimizes collateral damage to normal neural elements. This precision reduces the likelihood of adverse neurological side effects, which are a significant concern in current GBM regimens and contribute to the poor treatment tolerance among patients.
While these preclinical findings are highly encouraging, the transition from bench to bedside involves a rigorous pathway. The molecule must undergo further optimization to enhance its pharmacokinetics and pharmacodynamics, ensuring efficacy and safety in human subjects. Subsequent phases will require exhaustive clinical trials to evaluate dosing, therapeutic benefit, and long-term risks before potential approval by regulatory bodies such as the U.S. Food and Drug Administration.
Dr. Li underscored the novelty of this approach, stating that it exploits a biological pathway previously untargeted in glioblastoma therapy. By focusing on a critical dependency unique to GBM cells, this inhibitor exemplifies a precision medicine strategy designed to circumvent the limitations of generic cytotoxic treatments. If successful, this therapy could revolutionize clinical management of glioblastoma, offering patients a treatment that meaningfully extends survival and preserves neurological function.
The research was bolstered by the National Institutes of Health and foundations committed to cancer innovation, highlighting not only the scientific significance but the collaborative funding essential in tackling such a formidable disease. Furthermore, the establishment of AVIL Therapeutics by Dr. Li represents a translational effort to expedite the development of AVIL inhibitors toward clinical application, bridging the gap between scientific discovery and patient care.
The broader implications extend beyond glioblastoma, as the mechanistic insights into cytoskeletal regulation and oncogene function could illuminate therapeutic strategies for other refractory cancers. Targeting tumor-specific molecular aberrations with finely tuned small molecules invites a paradigm shift, moving away from blanket cytotoxicity toward tailored intervention at the heart of cancer cell survival mechanisms.
Glioblastoma’s dire prognosis and the unchanged standard of care over decades have fueled patient desperation and the medical community’s commitment to innovation. This discovery embodies hope by delivering a scientifically informed, mechanistically precise, and patient-friendly treatment modality. The advent of an orally administered pill that can discriminatorily annihilate glioblastoma cells, sparing healthy brain tissue, symbolizes a milestone in oncology and neurology alike.
The ongoing work to refine and bring this AVIL inhibitor into human trials reflects a broader imperative: translating molecular oncology insights into tangible, life-saving therapies. As research advances, it holds promise not only for the thousands diagnosed annually with glioblastoma but also underscores the transformative potential of precision-targeted cancer therapeutics in modern medicine.
Subject of Research: Glioblastoma molecular mechanisms and targeted therapy development.
Article Title: Not provided.
News Publication Date: Not explicitly stated in the source.
Web References:
https://dx.doi.org/10.1126/scitranslmed.adt1211
http://makingofmedicine.virginia.edu/
References:
Li, H., Xie, Z., Janczyk, P. Ł., et al. (published in Science Translational Medicine)
Image Credits: UVA Health
Keywords: Brain cancer, Glioblastomas, Diseases and disorders, Cancer, Clinical medicine, Medical treatments, Cancer treatments, Health and medicine, Life sciences, Cell biology, Cells, Cancer cells, Glioblastoma cells
Tags: AVIL gene and glioblastomachallenges in glioblastoma treatmentDr. Hui Li glioblastoma studyglioblastoma prognosis and survival ratesglioblastoma treatment advancementsinnovative therapies for aggressive brain cancerinvasive nature of glioblastoma tumorsmolecular targets in glioblastoma therapysmall molecule inhibitors for brain cancertargeted therapies for glioblastoma multiformetherapeutic strategies for brain cancerUniversity of Virginia cancer research