In the relentless pursuit of innovative cancer therapies, the cGAS–STING (cyclic GMP-AMP synthase–stimulator of interferon genes) pathway has emerged as a beacon of hope within the realm of immuno-oncology. Traditionally celebrated for its role in orchestrating immune responses via interferon induction, recent groundbreaking research has revealed a novel dimension of STING agonists that extends far beyond immune modulation. Investigators have discovered that these agonists exert a direct cytotoxic effect on tumors of the nervous system by harnessing an intrinsic signaling axis within malignant cells themselves. This revelation redefines our understanding of STING biology, unveiling a tumor-intrinsic mechanism that culminates in the expression of HMGN2—a chromatin-bound protein pivotal to apoptotic induction.
The therapeutic landscape surrounding STING agonists has long been characterized by a paradox: potent preclinical efficacy often fails to translate into consistent clinical success. The heterogeneity in tumor responsiveness has perplexed oncologists and researchers alike, prompting a rigorous examination of intra-tumoral molecular circuits that might elucidate these disparities. Predominantly, prior focus gravitated toward STING’s modulation of the tumor immune microenvironment, sometimes neglecting the autonomous tumor-cell-intrinsic pathways that potentially dictate sensitivity or resistance to these agonists.
A collaborative team of scientists spanning several prestigious Chinese institutions—including the First Medical Center of the Chinese PLA General Hospital and Peking University School of Life Sciences—sought to decode this enigma at a molecular resolution. Their seminal study, published in the renowned journal Cancer Biology & Medicine in late 2025, meticulously delineates how STING activation precipitates a STAT1-dependent transcriptional cascade culminating in the upregulation of HMGN2, which in turn orchestrates tumor cell apoptosis. These findings spotlight the critical role of tumor-intrinsic signaling cascades in mediating therapeutic outcomes and offer a mechanistic framework for leveraging STING agonists with newfound precision.
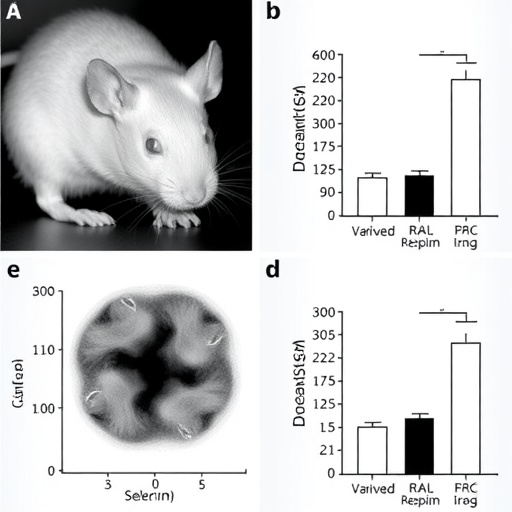
Experimental interrogation began with a comprehensive survey of diverse tumor cell lines to gauge their susceptibility to STING agonist treatment. Strikingly, neural-origin tumors—specifically neuroblastoma and glioblastoma—exhibited robust proliferative inhibition upon exposure to the synthetic STING agonist SR-717. Contrastingly, several non-neural carcinomas demonstrated scant response, underscoring the tissue-specific nature of this effect. Furthermore, murine xenograft models recapitulated these results, even under immunodeficient conditions, suggesting that the antitumor activity transcends conventional immune cell engagement and hinges upon intrinsic tumor cell pathways.
To unravel the molecular architecture underlying this phenomenon, the researchers deployed high-throughput RNA sequencing, which unveiled a constellation of differentially expressed genes post-STING activation. Central among these was HMGN2, a nucleosome-binding protein previously implicated in chromatin remodeling but now unveiled as a linchpin in apoptotic execution. Functional assays confirmed that overexpressing HMGN2 in tumor cells independently triggered apoptosis, whereas CRISPR-mediated HMGN2 knockout abrogated the antitumor efficacy of STING agonists, decisively establishing causality.
The axis linking STING activation and HMGN2 transcription was further explored through mechanistic studies revealing the indispensable role of STAT1, a well-characterized transcription factor commonly downstream of interferon signaling. Chromatin immunoprecipitation assays elegantly demonstrated STAT1’s direct binding to promoter regions of HMGN2, forming a previously uncharacterized STING–STAT1–HMGN2 signaling axis. This pathway operates as an autonomous molecular conduit within tumor cells, effecting apoptotic programs that culminate in tumor suppression independent of canonical immune responses.
Beyond mechanistic insights, bioinformatic analyses leveraging publicly accessible oncologic datasets reinforced the clinical relevance of HMGN2. Elevated HMGN2 transcript levels correlated positively with improved patient survival across various malignancies, positioning this protein not only as a therapeutic mediator but also as a valuable prognostic biomarker. This dual role opens the tantalizing prospect of employing HMGN2 expression as a stratification tool to identify patients most apt to benefit from STING-targeted interventions.
The implications of this research resonate deeply within the field of precision oncology. Historically, the application of STING agonists has been hamstrung by unpredictable patient responses and an absence of reliable biomarkers. By shifting the paradigm to acknowledge tumor cell-intrinsic regulation as a determinant of efficacy, clinicians may soon tailor treatments guided by molecular profiling of the STING–STAT1–HMGN2 axis. Such stratification promises to optimize therapeutic indices, maximize patient benefit, and reduce unwarranted exposure to ineffective regimens.
The neuro-oncology domain stands to gain substantially from these findings. Given the aggressive nature and immunosuppressive microenvironment characteristic of nervous system tumors such as glioblastoma, therapeutic strategies that circumvent immune evasion mechanisms are critically needed. STING agonists capable of directly engaging tumor-intrinsic apoptotic pathways may represent a paradigm shift, offering a novel modality to target these recalcitrant malignancies with enhanced specificity and potency.
Looking forward, this discovery invites exploration into combination therapies designed to potentiate HMGN2 expression or restore the sensitivity of resistant tumors to STING modulation. Agents that synergize with STAT1 activation or chromatin remodeling may amplify apoptotic cascades, thereby broadening the therapeutic window. Additionally, the universality of the STING–STAT1–HMGN2 axis suggests potential applicability beyond neural tumors, warranting investigation across diverse cancer types where STING pathway engagement remains underexploited.
This transformative insight into the molecular determinants governing tumor responses to STING agonists underscores the evolution of cancer immunotherapy from a primarily immune-centric strategy to an integrated approach encompassing direct tumor cell targeting. It exemplifies how dissecting intracellular signaling networks can reveal critical vulnerabilities previously masked by the complexity of tumor-immune interactions. Ultimately, these advances herald a future wherein STING agonists transcend their experimental roots to become tailored, mechanism-driven weapons in the oncologist’s arsenal.
As the clinical community wrestles with the formidable challenge of durable, effective cancer treatments, the STING–STAT1–HMGN2 axis emerges as a beacon illuminating new therapeutic and diagnostic horizons. By harnessing the intrinsic apoptotic machinery within tumor cells, this pathway offers a compelling template for the next generation of immunotherapeutic innovations. The journey from bench to bedside is poised to leverage these findings, translating molecular discovery into tangible clinical impact with the promise of improved patient outcomes.
Subject of Research: Not applicable
Article Title: Antitumor effects of STING agonists on nervous system tumors via tumor-intrinsic STING-STAT1-mediated HMGN2 expression
News Publication Date: 30-Oct-2025
References:
DOI: 10.20892/j.issn.2095-3941.2025.0326
Image Credits: Cancer Biology & Medicine
Keywords: Agonists, STING pathway, STAT1, HMGN2, tumor-intrinsic signaling, nervous system tumors, neuroblastoma, glioblastoma, apoptosis, immunotherapy, cancer biomarkers, precision oncology
Tags: apoptotic induction in malignant cellscGAS–STING pathway in brain tumorschallenges in STING-based cancer therapydirect tumor cell targeting strategiesHMGN2 protein role in apoptosisimmune modulation in nervous system tumorsimmuno-oncology advances in brain cancermolecular mechanisms of brain tumor therapymolecular self-destruct mechanisms in tumorsSTING agonists cytotoxic effectstumor heterogeneity and treatment resistancetumor-intrinsic signaling in cancer