“In this study, we investigated the therapeutic effects and the underlying mechanisms of TAS-102 in combination with regorafenib against gastrointestinal cancers.”
Credit: 2024 Zhang et al.
“In this study, we investigated the therapeutic effects and the underlying mechanisms of TAS-102 in combination with regorafenib against gastrointestinal cancers.”
BUFFALO, NY- July 9, 2024 – A new research paper was published in Oncotarget’s Volume 15 on July 2, 2024, entitled, “Regorafenib synergizes with TAS102 against multiple gastrointestinal cancers and overcomes cancer stemness, trifluridine-induced angiogenesis, ERK1/2 and STAT3 signaling regardless of KRAS or BRAF mutational status.”
Single-agent TAS102 (trifluridine/tipiracil) and regorafenib are FDA-approved treatments for metastatic colorectal cancer (mCRC). Researchers previously reported that regorafenib combined with a fluoropyrimidine can delay disease progression in clinical case reports of multidrug-resistant mCRC patients. In this new study, researchers Jun Zhang, Lanlan Zhou, Shuai Zhao, and Wafik S. El-Deiry from Fox Chase Cancer Center and Brown University hypothesized that the combination of TAS102 and regorafenib may be active in CRC and other gastrointestinal (GI) cancers and may in the future provide a treatment option for patients with advanced GI cancer.
“We investigated the therapeutic effect of TAS102 in combination with regorafenib in preclinical studies employing cell culture, colonosphere assays that enrich for cancer stem cells, and in vivo.”
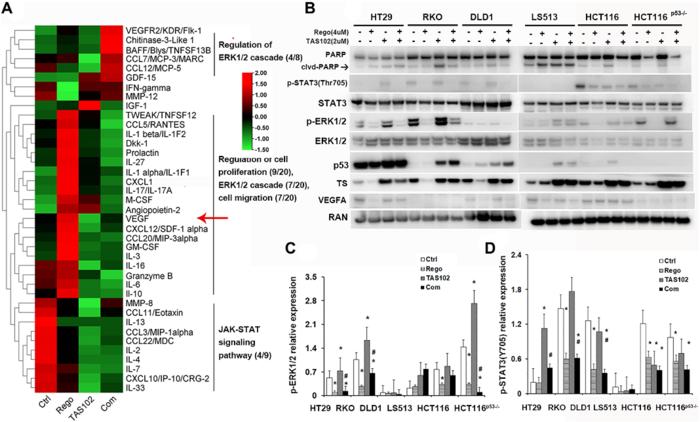
TAS102 in combination with regorafenib has synergistic activity against multiple GI cancers in vitro including colorectal and gastric cancer, but not liver cancer cells. TAS102 inhibits colonosphere formation and this effect is potentiated by regorafenib. In vivo anti-tumor effects of TAS102 plus regorafenib appear to be due to anti-proliferative effects, necrosis and angiogenesis inhibition.
Growth inhibition by TAS102 plus regorafenib occurs in xenografted tumors regardless of p53, KRAS or BRAF mutations, although more potent tumor suppression was observed with wild-type p53. Regorafenib significantly inhibits TAS102-induced angiogenesis and microvessel density in xenografted tumors, as well inhibits TAS102-induced ERK1/2 activation regardless of RAS or BRAF status in vivo. TAS102 plus regorafenib is a synergistic drug combination in preclinical models of GI cancer, with regorafenib suppressing TAS102-induced increase in microvessel density and p-ERK as contributing mechanisms.
“The TAS102 plus regorafenib drug combination may be further tested in gastric and other GI cancers.”
Continue reading: DOI: https://doi.org/10.18632/oncotarget.28602
Correspondence to: Wafik S. El-Deiry
Email: [email protected]
Keywords: TAS102, regorafenib, ERK1/2, angiogenesis, microvessel density
Click here to sign up for free Altmetric alerts about this article.
About Oncotarget: Oncotarget (a primarily oncology-focused, peer-reviewed, open access journal) aims to maximize research impact through insightful peer-review; eliminate borders between specialties by linking different fields of oncology, cancer research and biomedical sciences; and foster application of basic and clinical science.
Oncotarget is indexed and archived by PubMed/Medline, PubMed Central, Scopus, EMBASE, META (Chan Zuckerberg Initiative) (2018-2022), and Dimensions (Digital Science).
To learn more about Oncotarget, visit Oncotarget.com and connect with us on social media:
- X, formerly Twitter
- YouTube
- Spotify, and available wherever you listen to podcasts
Click here to subscribe to Oncotarget publication updates.
For media inquiries, please contact [email protected].
Oncotarget Journal Office
6666 East Quaker Street., Suite 1
Orchard Park, NY 14127
Phone: 1-800-922-0957 (option 2)
###
Journal
Oncotarget
DOI
10.18632/oncotarget.28602
Method of Research
Experimental study
Subject of Research
People
Article Title
Regorafenib synergizes with TAS102 against multiple gastrointestinal cancers and overcomes cancer stemness, trifluridine-induced angiogenesis, ERK1/2 and STAT3 signaling regardless of KRAS or BRAF mutational status
Article Publication Date
2-Jul-2024



