Recent advances in understanding autoimmune mechanisms have led to groundbreaking findings in Sjögren’s disease, a chronic condition primarily characterized by diminished salivary and lacrimal gland function. A new study, published in Experimental & Molecular Medicine in March 2026, sheds light on a critical molecular pathway involving NRIP1, ERα signaling, and MYC-driven suppression of AQP5, which collectively contribute to salivary gland dysfunction in this debilitating disorder. These discoveries not only deepen our knowledge of the disease’s molecular pathology but also point toward potential targeted therapies.
Sjögren’s disease is a multifaceted autoimmune disorder where the immune system erroneously attacks exocrine glands, particularly those responsible for producing saliva and tears. The resultant glandular hypofunction manifests clinically as dry mouth and dry eyes, symptoms that significantly impair quality of life. Despite extensive research, the precise intracellular molecular interactions mediating glandular impairment remained elusive until now. This study pioneers the elucidation of how NRIP1-mediated interference with estrogen receptor alpha (ERα) signaling triggers pathological changes leading to glandular insufficiency.
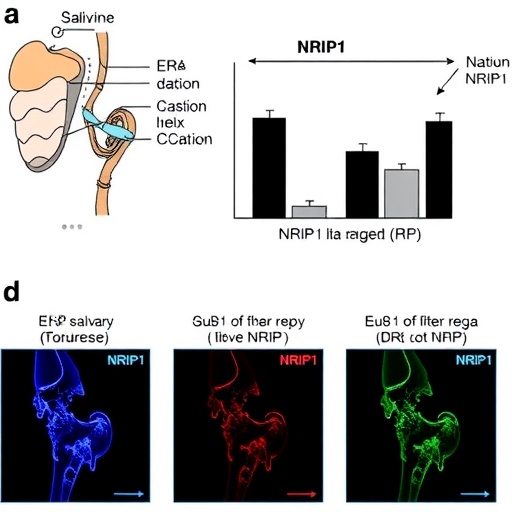
The estrogen receptor alpha (ERα) pathway has long been implicated in immune regulation and tissue homeostasis. Its role in salivary gland physiology is equally pivotal, as ERα signaling helps maintain proper glandular secretory function. Investigators identified NRIP1 (Nuclear Receptor Interacting Protein 1) as a disruptor of this pathway in Sjögren’s disease contexts. NRIP1 acts as a co-repressor, interfacing with ERα and dampening its transcriptional activity. This novel insight establishes a foundational mechanism by which hormonal signaling is subverted in the disease state.
Crucially, the study demonstrated that NRIP1 activation results in the suppression of Aquaporin 5 (AQP5), a water channel protein integral to saliva secretion. AQP5 facilitates water transport across salivary gland epithelial membranes, enabling fluid secretion critical for saliva production. The downregulation of AQP5 via NRIP1-mediated ERα disruption therefore directly compromises glandular output. This revelation bridges a crucial gap, linking receptor signaling defects to the mechanical failure of salivary secretion observed clinically.
Additionally, the research highlights a previously unappreciated role for the MYC oncogene in Sjögren’s disease-related salivary dysfunction. MYC, a well-characterized transcription factor renowned for its roles in proliferation and metabolism, was found to drive downstream pathological changes subsequent to NRIP1 and ERα pathway perturbations. The investigation delineates how aberrant MYC activation exacerbates transcriptional circuits that culminate in glandular decline, suggesting a multi-tiered mechanism of disease progression.
The interplay of NRIP1, ERα, and MYC delineated in this study crystallizes a complex molecular axis where hormonal signaling interfaces with gene regulation to govern glandular health. Importantly, the dynamic suppression of AQP5 by these factors encapsulates a convergent mechanism that translates molecular dysregulation directly into functional impairment. The data also suggest that intervening at nodes of this axis could restore signaling balance and potentially reverse or halt the progression of glandular dysfunction.
Methodologically, the study utilized a combination of patient-derived salivary gland tissue analyses, in vitro modeling, and sophisticated molecular biology techniques including chromatin immunoprecipitation sequencing (ChIP-seq) and transcriptomic profiling. This multifaceted experimental approach allowed the researchers to map signal transduction events with high precision and verify the causal relationships between NRIP1, ERα signaling disruption, AQP5 expression, and MYC responsiveness.
These findings hold profound implications for the broader field of autoimmune disease therapeutics. Targeting NRIP1 with selective modulators or reactivating ERα signaling pharmacologically may open therapeutic avenues previously unexplored. Moreover, strategies to modulate MYC activity could serve as adjunct treatments to protect glandular function, underscoring the value of mechanistic research in identifying therapeutic targets.
Given the chronic nature of Sjögren’s disease and its progressive impact on patient well-being, these insights demand attention from clinical researchers and pharmaceutical developers alike. Personalized treatment regimens that address molecular drivers such as NRIP1 and MYC could revolutionize management paradigms. Furthermore, the molecular markers identified may assist in earlier diagnosis, enabling interventions before irreversible glandular damage occurs.
This study also prompts further inquiry into how sex hormones influence autoimmune disease susceptibility and progression, especially since ERα is tightly linked to estrogen signaling. Understanding sex-related differences in Sjögren’s disease prevalence and severity through the lens of NRIP1 and ERα dynamics may uncover gender-specific therapeutic strategies. Such precision medicine approaches could dramatically improve outcomes for affected individuals.
In addition to its clinical ramifications, the research enriches fundamental biological knowledge regarding nuclear receptor co-regulators and transcription factor networks in glandular tissues. The NRIP1-ERα-MYC axis represents a compelling example of how transcriptional regulation orchestrates not only development and homeostasis but also pathological disarray in disease settings. These insights have relevance that extends beyond Sjögren’s disease, potentially informing studies of other hormone-sensitive autoimmune disorders.
Future studies building on these findings may explore how environmental factors or genetic predispositions modulate NRIP1 expression or ERα activity in salivary glands. Elucidating upstream regulators of this pathway could identify points of vulnerability for intervention or prevention. Furthermore, in vivo modeling to test candidate therapies targeting these molecular nodes will be critical to translating bench discoveries into clinical benefit.
Ultimately, the work by Chen, Pathak, Qin, and colleagues represents a landmark in molecular medicine, offering a coherent mechanistic framework to understand and combat salivary gland dysfunction in Sjögren’s disease. The integration of endocrinology, immunology, and transcriptional biology in their approach exemplifies the multidisciplinary synergy required to unravel complex autoimmune pathologies.
As the medical community grapples with autoimmune diseases’ intricate etiologies, studies like this herald a new era of targeted, mechanism-based therapies. The potential to restore gland function and alleviate chronic symptoms offers hope to millions suffering from Sjögren’s disease and related disorders. Above all, the elegant demonstration of NRIP1’s disruptive role on ERα signaling and downstream molecular cascades provides a compelling target for future clinical innovation.
The publication underscores the power of combining molecular insights with clinical relevance to forge paths toward precision medicine. By catching the molecular “fault lines” underlying glandular dysfunction, researchers can develop interventions tailored to disease-specific mechanisms rather than merely managing symptoms, fundamentally altering patient care.
With continued research and investment, these findings may soon translate into tangible benefits for patients living with Sjögren’s disease, turning molecular discoveries into real-world healing. The path toward improved quality of life for those affected now seems clearer than ever, propelled by this pivotal research uncovering the molecular choreography of salivary gland impairment.
Subject of Research: Molecular mechanisms underlying salivary gland dysfunction in Sjögren’s disease, focusing on NRIP1’s disruption of ERα signaling and MYC-driven pathways.
Article Title: NRIP1 disrupts ERα signal in Sjögren’s disease via AQP5 suppression and MYC-driven salivary dysfunction.
Article References:
Chen, B., Pathak, J.L., Qin, X. et al. NRIP1 disrupts ERα signal in Sjögren’s disease via AQP5 suppression and MYC-driven salivary dysfunction. Exp Mol Med (2026). https://doi.org/10.1038/s12276-026-01671-w
Image Credits: AI Generated
DOI: 10.1038/s12276-026-01671-w (Published 13 March 2026)
Tags: autoimmune exocrine gland damageautoimmune salivary gland dysfunctionERα signaling in salivary glandsestrogen receptor alpha in immune regulationintracellular pathways in glandular hypofunctionmolecular pathology of Sjögren’s diseaseMYC-driven AQP5 suppressionNRIP1 and estrogen receptor interactionNRIP1 role in autoimmune diseasesalivary gland secretory dysfunctionSjögren’s syndrome molecular mechanismstargeted therapies for Sjögren’s