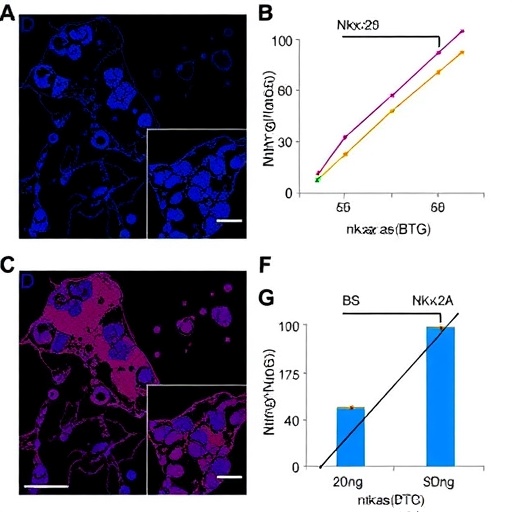
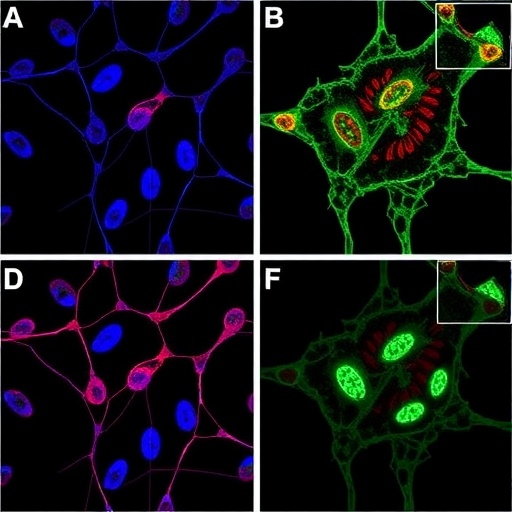
In a groundbreaking study poised to reshape our understanding of breast cancer biology, researchers have uncovered a novel regulatory axis involving long non-coding RNA Nkx2-2as and the BTG2 gene that impedes breast cancer progression by modulating the pivotal Wnt/β-catenin signaling pathway. This intricate molecular interplay shines a spotlight on potential therapeutic targets, offering fresh hope for combating one of the most prevalent malignancies affecting women worldwide.
The Wnt/β-catenin pathway has long been recognized for its critical role in cellular development, proliferation, and oncogenesis; its dysregulation is associated with various cancers, including breast carcinoma. However, the mechanisms governing this pathway’s modulation, particularly through non-coding genomic elements, remain only partially elucidated. The study’s identification of the lncRNA Nkx2-2as as a suppressive regulator underscores the emerging significance of non-coding RNAs in cancer pathophysiology.
Long non-coding RNAs (lncRNAs) have risen from obscurity to the forefront of cancer research due to their versatile roles in gene expression regulation, chromatin remodeling, and signaling cascades. Unlike protein-coding genes, lncRNAs do not translate into proteins but exert their regulatory influence via diverse mechanisms, including acting as molecular sponges, scaffolds, or guides for transcription factors. The revelation that Nkx2-2as functions as a crucial modulator of the Wnt pathway adds a vital piece to the intricate puzzle of breast cancer molecular dynamics.
Central to this regulatory axis is BTG2 (B-cell translocation gene 2), a well-characterized tumor suppressor known for its antiproliferative effects and involvement in cell cycle regulation. The study illuminates how Nkx2-2as positively influences BTG2 expression, which in turn represses canonical Wnt signaling components, thereby thwarting oncogenic signaling cascades that promote tumor growth and metastasis. This cascade represents a finely tuned molecular switch balancing cellular homeostasis and malignant transformation.
The researchers utilized a comprehensive suite of molecular biology techniques, including quantitative PCR, Western blotting, and RNA interference, to dissect the functional relationship between Nkx2-2as, BTG2, and the Wnt pathway. Their data demonstrated that silencing Nkx2-2as resulted in diminished BTG2 levels and concomitant activation of β-catenin, a transcriptionally active protein that orchestrates the expression of genes driving proliferation and invasion in breast cancer cells.
Furthermore, patient-derived breast tumor specimens exhibited significantly reduced Nkx2-2as and BTG2 expression compared to normal breast tissue, correlating inversely with markers of poor prognosis such as high tumor grade and metastasis. These clinical associations validate the biological relevance of the Nkx2-2as/BTG2 axis and underscore its potential utility as a prognostic biomarker for aggressive breast cancer phenotypes.
The implications of modulating the Nkx2-2as/BTG2 pathway extend beyond prognostication, opening avenues for novel therapeutic interventions. Strategies to restore or mimic Nkx2-2as function could potentially reinstate BTG2-mediated repression of Wnt/β-catenin signaling, stymieing tumor progression. Such approaches may include synthetic lncRNA delivery, small molecule activators, or gene editing technologies that specifically enhance the expression or stability of Nkx2-2as.
Interestingly, the study also delves into the downstream effectors of BTG2, highlighting its role in destabilizing β-catenin via ubiquitination and proteasomal degradation. This post-translational regulatory mechanism is crucial in maintaining controlled Wnt signaling and preventing aberrant activation that fuels oncogenesis. By substantiating BTG2’s involvement in these intricate cellular processes, the research provides a molecular rationale for its tumor suppressive capacity.
The dynamic microenvironment of breast tumors is a complex milieu where signaling pathways intercross, creating redundancies and feedback loops that challenge therapeutic targeting. The discovery of the Nkx2-2as/BTG2 axis adds an essential component to this network, emphasizing how non-coding elements orchestrate critical checkpoints in cancer progression. This knowledge enriches our comprehension of tumor heterogeneity and may guide the development of combination therapies targeting multiple nodes within the oncogenic circuit.
In addition to molecular insights, the research acknowledges the translational potential of their findings. Clinical trials utilizing Wnt inhibitors have been hamstrung by limited efficacy and toxicities stemming from the pathway’s pervasive role in normal tissue homeostasis. Modulating the pathway indirectly via lncRNA regulation offers a subtler, potentially less toxic approach by exploiting natural cellular safeguards such as BTG2.
As precision medicine continues to evolve, integrating lncRNA profiles into patient stratification protocols could enhance treatment personalization. For example, patients exhibiting low Nkx2-2as and BTG2 expression might be identified as candidates for lncRNA-targeted therapies or experimental agents aimed at reinstating tumor suppressive networks. This tailored strategy could improve outcomes and reduce the burden of broad-spectrum cytotoxic therapies.
Further research is warranted to elucidate the upstream regulators controlling Nkx2-2as expression and stability. Epigenetic modifications, transcription factor binding, and microRNA interactions may converge to modulate this lncRNA’s availability, presenting additional targets for intervention. Comprehensive mapping of these regulatory layers will refine our understanding of breast cancer biology and therapeutic vulnerabilities.
Moreover, the interplay between Nkx2-2as/BTG2 and other signaling pathways, such as PI3K/AKT or Notch, remains to be fully explored. Crosstalk among oncogenic circuits often dictates tumor behavior and resistance patterns; thus, dissecting these relationships could reveal synergistic targets and inform combinatorial regimens designed to thwart adaptive tumor escape mechanisms.
This study’s revelations underscore the paradigm shift towards appreciating the non-coding genome’s profound impact on cancer. Beyond the canonical protein-coding genes, the vast landscape of lncRNAs represents a treasure trove of regulatory elements intricately woven into cancer’s molecular fabric. As technologies advance to probe this complexity, fresh opportunities arise for diagnostic, prognostic, and therapeutic innovations.
In sum, the identification of the long non-coding RNA Nkx2-2as as a critical modulator of BTG2 expression and Wnt/β-catenin signaling provides a compelling narrative linking non-coding RNA biology to breast cancer progression. This axis not only deepens our molecular understanding but also heralds a promising frontier for novel therapeutic intervention, potentially altering the trajectory of breast cancer management in the years ahead.
Subject of Research: The regulatory role of long non-coding RNA Nkx2-2as and BTG2 in breast cancer progression through modulation of Wnt/β-catenin signaling.
Article Title: Long non-coding RNA Nkx2-2as/BTG2 axis attenuates breast cancer progression by targeting Wnt/β-catenin signaling.
Article References:
Ravi, A.K., Muthukrishnan, S., Gunasangkaran, G. et al. Long non-coding RNA Nkx2-2as/BTG2 axis attenuates breast cancer progression by targeting Wnt/β-catenin signaling. Med Oncol 43, 12 (2026). https://doi.org/10.1007/s12032-025-03141-1
Image Credits: AI Generated
DOI: https://doi.org/10.1007/s12032-025-03141-1
Tags: breast cancer progression suppressionBTG2 gene breast cancercancer biology research breakthroughschromatin remodeling in oncologyemerging roles of lncRNAsgene expression regulation in cancerlncRNA regulatory mechanismsmolecular mechanisms of tumor suppressionNkx2-2as long non-coding RNAnon-coding RNAs in oncologytherapeutic targets in breast cancerWnt beta-catenin signaling pathway