Ovarian cancer remains the deadliest gynecological malignancy, primarily due to its stealthy progression and late-stage diagnosis. Unlike many cancers that metastasize through the bloodstream, ovarian cancer disseminates aggressively within the abdominal cavity, eluding early detection and presenting profound treatment challenges. Recent groundbreaking research from Nagoya University, published in Science Advances, has uncovered a pivotal mechanism behind this rapid intra-abdominal spread: ovarian cancer cells co-opt mesothelial cells lining the peritoneal cavity to actively invade tissues and resist chemotherapy.
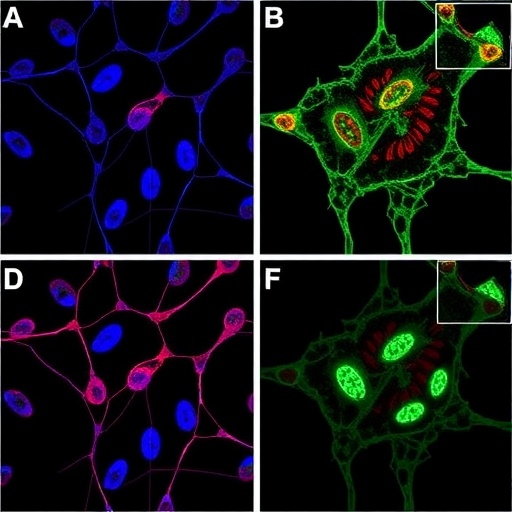
This study reveals a sophisticated cellular partnership wherein ovarian cancer cells recruit mesothelial cells to join them in hybrid spherical clusters within the ascitic fluid. These mesothelial cells, normally responsible for protecting and lining the abdominal organs, undergo a transformation upon exposure to a cancer-secreted protein called TGF-β1. This transformation enables the mesothelial cells to develop specialized, finger-like protrusions known as invadopodia that mechanically breach surrounding tissues, effectively clearing invasion paths for the cancer cells.
Distinct from tumors such as breast or lung cancer that metastasize via vascular routes, ovarian cancer cells exploit the dynamic environment of the peritoneal cavity, where fluid movement facilitates cellular dispersal. Floating freely in ascitic fluid, ovarian cancer cells encounter shed mesothelial cells, and through a process of cellular adhesion and molecular signaling, they form tightly bound hybrid spheroids. Approximately 60% of such cancer spheres contain these recruited mesothelial cells, illustrating the prevalence and importance of this interaction in cancer progression.
Intriguingly, the cancer cells themselves remain relatively genetically stable during this metastatic journey, relying instead on the mesothelial cells to perform the “heavy lifting” of tissue invasion. By outsourcing the mechanical work to their mesothelial partners, cancer cells maintain a minimal level of molecular alterations, merely following the invasion routes sculpted by the invadopodia. This strategy not only facilitates rapid tissue penetration but also enhances the clusters’ survival, as the hybrid spheroids exhibit marked resistance to standard chemotherapy agents.
The researchers employed advanced live-cell microscopic imaging techniques to observe these cellular behaviors within fluid samples obtained from ovarian cancer patients. This real-time visualization provided direct evidence of mesothelial cell recruitment, spheroid formation, and the active tissue invasion carried out by invadopodia structures. Complementary experiments in murine models and single-cell transcriptomic profiling further validated the human relevance and molecular underpinnings of these findings.
Dr. Kaname Uno, the study’s lead author, highlights that the identification of this hybrid cell strategy unravels a novel dimension of tumor biology. Previously, the floating stage of ovarian cancer cells within the abdomen represented a black box—cancer’s elusive tactic to evade immune surveillance and therapeutic regimes. Understanding that mesothelial cells are complicit in fostering both invasion and chemoresistance opens transformative possibilities for clinical interventions.
The biology of invadopodia has long intrigued cancer scientists due to their role in matrix degradation and invasion. This study extends that knowledge by illustrating mesothelial cells, traditionally viewed as passive bystanders or barriers, as active accomplices remodeled by cancer signals. The invocation of TGF-β1 signaling as the molecular switch manipulating mesothelial cell behavior provides a tangible drug target. Inhibitors of this signaling pathway may disrupt the formation of these dangerous hybrid invasions, thereby reducing metastatic spread and improving chemotherapy efficacy.
Furthermore, this discovery suggests a new biomarker strategy: detection and monitoring of these hybrid spheroids in patient abdominal fluid could become a proxy indicator of disease progression and treatment response. Unlike blood-based markers, which may be less predictive in ovarian cancer’s unique metastatic context, analyzing peritoneal fluid may offer better prognostic value and guide personalized therapeutic decisions.
The implication of these findings transcends ovarian cancer. They hint at broader paradigms in cancer metastasis where tumor cells may recruit and co-opt non-malignant stromal or protective cells to facilitate invasion and survival. This concept opens fresh avenues for research into other cancers that spread via body cavity fluids, challenging researchers to rethink traditional models focused solely on cancer cell-autonomous behaviors.
Dr. Uno’s transition from clinical gynecology to cancer research imparts a poignant undercurrent to this study. Motivated by the tragic loss of a patient whose ovarian cancer progressed too swiftly for early diagnosis, he pursued scientific inquiry that now lays groundwork for earlier detection and innovative treatments. The human element behind this work underscores the urgent need for better understanding and combatting ovarian cancer’s deadly progression.
In summary, the study from Nagoya University elucidates a previously unrecognized cellular collaboration that accelerates ovarian cancer metastasis through the abdomen. By hijacking protective mesothelial cells to forge invasive spheroids, ovarian cancer cells gain both a physical advantage in tissue invasion and a biochemical shield against chemotherapy. This advances our understanding of peritoneal metastasis and sets the stage for novel therapeutic targets that disrupt this malignant alliance.
The future of ovarian cancer treatment may lie in targeting these hybrid clusters, particularly by blocking the TGF-β1 induced mesothelial transformation and invadopodia development. Such strategies promise not only to hinder the cancer’s invasive march but also to enhance patients’ responsiveness to existing chemotherapy regimens. Continued research in this groundbreaking direction could significantly shift the landscape in managing one of the most lethal women’s cancers.
Subject of Research: Human tissue samples
Article Title: Mesothelial cells promote peritoneal invasion and metastasis of ascites-derived ovarian cancer cells through spheroid formation
News Publication Date: 6-Feb-2026
Web References:
https://doi.org/10.1126/sciadv.adu5944
References:
Uno et al., 2026
Image Credits:
Uno et al., 2026
Keywords:
Ovarian cancer, mesothelial cells, peritoneal metastasis, hybrid spheroids, invadopodia, TGF-β1 signaling, ascitic fluid, chemotherapy resistance, cancer invasion, cellular cooperation, tumor microenvironment, metastatic mechanisms
Tags: abdominal cancer spread researchascitic fluid and cancer cell behaviorcancer treatment challenges in gynecologychemotherapy resistance in ovarian cancerhybrid cellular clusters in cancerinvasive cancer cell behaviorlate-stage ovarian cancer diagnosismesothelial cell role in cancerovarian cancer and mesothelial cell interactionovarian cancer metastasis mechanismsperitoneal cavity cancer dynamicsTGF-β1 protein impact on cells