Triple-negative breast cancer (TNBC) is notoriously challenging to treat, primarily because nearly half of all patients eventually develop resistance to standard therapies. This therapeutic resistance not only limits the effectiveness of treatment but also significantly worsens patient outcomes by increasing the likelihood of tumor recurrence. In a pioneering study conducted at Baylor College of Medicine, researchers have unveiled that TNBC does not rely on a single mechanism to become resistant; rather, distinct and mutually exclusive pathways operate depending on the cellular makeup of the tumor. This breakthrough finding offers a promising avenue to tailor treatment strategies according to the tumor’s unique biology, potentially circumventing resistance and improving survival rates.
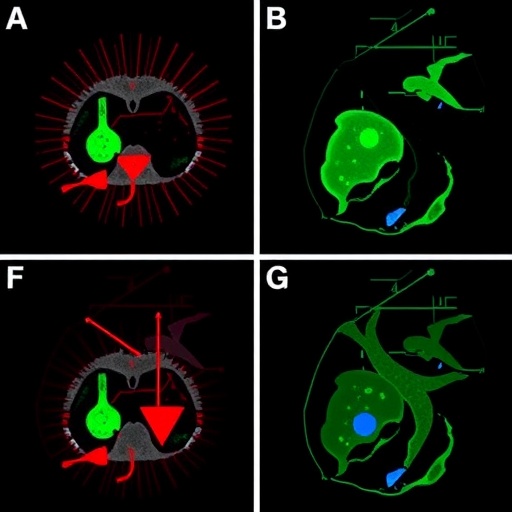
The team’s research, detailed in the Journal of Clinical Investigation, builds on prior observations that TNBC tumors are heterogeneous, exhibiting different cellular compositions that include diverse populations of tumor and immune cells. Among these immune cells, neutrophils and macrophages stand out as critical players. Dr. Xiang Zhang, the study’s senior author at Baylor College of Medicine, emphasized that variability in this immune cell landscape dictates the specific resistance mechanism a tumor might develop. Epithelial-like TNBC tumors, which harbor both macrophages and neutrophils, principally utilize neutrophil-driven pathways to evade chemotherapy, whereas mesenchymal-like tumors, dominated by macrophages, exploit these cells’ unique functional shifts to survive treatment.
Utilizing a robust experimental framework that combined investigations of human patient tissue samples with sophisticated mouse models, the researchers were able to dissect these resistance pathways with high resolution. Their previous research highlighted neutrophils as key facilitators in epithelial-like TNBC resistance, but the current study delves into mesenchymal-like tumors to examine macrophages’ role. Strikingly, macrophages in these tumors are not mere bystanders; chemotherapy itself appears to reprogram these immune cells, converting them from tumor-fighting sentinels into agents that actively suppress the immune response and assist tumor survival.
Dr. Liqun Yu, a co-first author of the study, elaborated that reprogrammed macrophages engage in a paradoxical behavior: while they continue to engulf and clear cancer cells, they simultaneously produce immune-suppressive molecules such as C1q and resolvin. These compounds inhibit cytotoxic immune cell activity, thereby creating an immunosuppressive microenvironment favorable for tumor cells to persist despite chemotherapy. This dual functionality of macrophages introduces a complex layer of resistance, highlighting the intricate crosstalk between cancer cells and the immune system within the tumor microenvironment.
To counteract this macrophage-mediated resistance, the research team explored several therapeutic strategies aimed at either eliminating macrophages or blocking their recruitment into mesenchymal-like tumors. Remarkably, these interventions restored sensitivity to chemotherapy, indicating that disabling the immunosuppressive capabilities of macrophages could reinvigorate the body’s natural defense mechanisms against tumor cells. Additionally, antagonizing the molecules secreted by the reprogrammed macrophages further revived the immune system’s antitumor response, underscoring new potential drug targets for overcoming resistance.
The implications of these findings extend beyond understanding resistance mechanisms; they usher in a new paradigm for personalized TNBC treatment. By profiling a tumor’s immune cell composition prior to therapy—discerning whether it aligns more closely with epithelial-like or mesenchymal-like characteristics—clinicians could predict the specific pathways the tumor might exploit to resist chemotherapy. This proactive approach could enable tailored treatment regimens that preempt resistance, employing immune modulatory agents alongside standard chemotherapy to block the tumor’s escape routes.
Furthermore, the delineation of discrete, mutually exclusive resistance mechanisms challenges the traditional one-size-fits-all model prevalent in oncological therapy. As noted by Dr. Zhang, the study reinforces that TNBC is not a monolithic entity; its heterogeneity demands nuanced therapeutic interventions. Targeting neutrophil-driven pathways in epithelial-like tumors while simultaneously addressing macrophage-mediated resistance in mesenchymal-like tumors could offer a dual-pronged strategy that enhances treatment efficacy across the TNBC spectrum.
The investigative team credits their progress to the integration of patient-derived samples for clinical relevance and genetically engineered mouse models that offer mechanistic insights. This translational research framework bridges laboratory findings with potential clinical applications, accelerating the pathway to novel therapies. Co-author Bo Wei from the University of Texas MD Anderson Cancer Center also contributed pivotal expertise, underscoring the collaborative, multi-institutional effort necessary to tackle the complexity of TNBC.
The study was supported through various grants from leading institutions including the National Institutes of Health, the Department of Defense, the National Cancer Institute, and the Breast Cancer Research Foundation. Such financial backing reflects the critical importance of continuing to unravel the cellular and molecular underpinnings of cancer resistance, which remains a substantial barrier to curing aggressive cancers like TNBC.
In summary, this groundbreaking research reveals that therapeutic resistance in TNBC emerges from distinct myeloid immune circuits shaped by the tumor’s cellular composition. The interplay of neutrophils and macrophages in crafting resistance pathways underscores the adaptive capabilities of the tumor microenvironment under therapeutic pressure. Targeting these circuits offers a tantalizing prospect for developing more effective, personalized therapies—potentially transforming the prognosis for patients afflicted by one of the most refractory breast cancer subtypes.
Subject of Research: Human tissue samples
Article Title: Inflammation- and resolution-programmed myeloid circuits govern therapeutic resistance in epithelial and mesenchymal triple-negative breast cancer
News Publication Date: 17-Feb-2026
Web References:
Journal of Clinical Investigation
Previous related study
References: DOI 10.1172/JCI198815
Keywords: Triple-negative breast cancer, TNBC, therapy resistance, macrophages, neutrophils, immunosuppression, tumor microenvironment, personalized medicine, chemotherapy resistance, immune modulation
Tags: cellular composition in tumor resistanceepithelial-like TNBC resistance mechanismsimmune cell influence on cancer therapy outcomesmacrophage involvement in TNBCmesenchymal-like TNBC pathwaysneutrophil-driven chemotherapy resistanceovercoming chemotherapy resistance in breast cancerpersonalized treatment for TNBCTNBC immune cell heterogeneityTNBC tumor recurrence factorstriple-negative breast cancer therapy resistancetumor microenvironment and therapy resistance