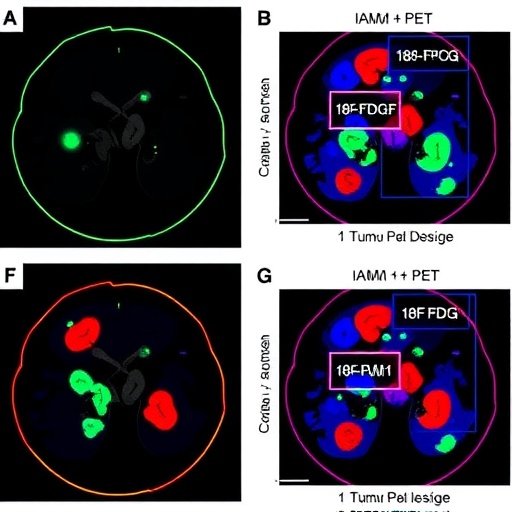
Radiotherapy (RT) remains a cornerstone in the clinical management of cancer, deployed in over half of all cancer patients worldwide. Despite its widespread application, the nuanced biological processes underlying tumor response to RT are incompletely understood. Advanced imaging modalities like positron emission tomography (PET) using the glucose analog ^18F-fluorodeoxyglucose (^18F-FDG) have revolutionized how clinicians noninvasively assess metabolic activity within tumors. Intriguingly, following RT, many tumors manifest transient surges in ^18F-FDG uptake—phenomena historically labeled as “metabolic flares.” These flares have conventionally been interpreted as localized inflammatory responses, yet the precise cellular and molecular drivers remained elusive.
In a breakthrough study published in Protein & Cell, researchers delved deeply into the mechanistic underpinnings of post-RT metabolic flares, integrating rigorous analyses of both human tumor specimens and sophisticated murine cancer models. Their findings pivotally identify the upregulation of intracellular adhesion molecule-1 (ICAM-1) in tumor tissues as a critical instigator of these PET imaging flare-ups. RT robustly boosts ICAM-1 expression within the tumor microenvironment, a process intricately linked with enhanced infiltration and clustering of tumor-resident T cells, effectively reprogramming their metabolic and functional states.
Genetic ablation experiments revealed that loss of ICAM-1 severely diminished the magnitude of RT-induced ^18F-FDG flares. This attenuation was traceable predominantly to a reduction in T cell glucose uptake and intratumoral accumulation, rather than fluctuations in myeloid cell activity. These data decisively shift the paradigm, elevating T cells—rather than mere inflammatory cells—as primary drivers of the metabolic signals detected by ^18F-FDG PET post-radiotherapy.
At the molecular signaling level, RT-induced ICAM-1 was shown to engage lymphocyte function-associated antigen-1 (LFA-1), one of its principal binding partners on T cells. This interaction orchestrates clustering and retention of T cells within tumors. Crucially, this ICAM-1/LFA-1 mediated adhesion event activates a downstream signaling cascade governed by the PI3K–AKT–mTOR axis, well known for its pivotal role in cellular metabolism and immune cell effector function.
Activation of the PI3K–AKT–mTOR pathway triggers profound metabolic rewiring in T cells, characterized by increased glycolysis and enhanced tricarboxylic acid (TCA) cycle flux. This metabolic reprogramming equips tumor-infiltrating T cells with the bioenergetic and biosynthetic capacity to sustain robust effector functions in the hostile tumor microenvironment. The elevated glucose metabolism of these T cells, visualized by heightened ^18F-FDG uptake, explains the observed PET flares post-radiotherapy and links metabolic imaging signals directly to adaptive immune dynamics.
This mechanistic insight fundamentally enriches the clinical interpretation of post-RT PET scans. Traditionally, increased ^18F-FDG signals have been confounded as either tumor progression or inflammatory pseudoprogression, complicating therapeutic decision-making. The identification of ICAM-1 as a marker of T cell metabolic engagement offers a novel biomarker to distinguish these distinct biological phenomena, fostering more precise and individualized treatment monitoring.
Importantly, these discoveries open promising avenues to harness metabolic imaging as a proxy for intratumoral immune activity, offering clinicians a window into antitumor immune responses elicited by RT. This could be pivotal for optimizing RT-based combination therapies, particularly those integrating immune checkpoint inhibitors or adoptive T cell transfer strategies, where timely assessment of immune activation dictates therapeutic success.
From a therapeutic development perspective, targeting the ICAM-1/LFA-1 axis or its downstream PI3K–AKT–mTOR signaling cascade may modulate T cell infiltration and function in tumors. Such interventions could synergize with RT to amplify antitumor immunity or circumvent resistance mechanisms. Moreover, metabolic modulation of T cells guided by this pathway provides a strategic focal point for designing novel immunometabolic therapies.
This study exemplifies an integrative systems biology approach, fusing tumor biology, immunology, metabolic analysis, and advanced imaging, underscoring the complex crosstalk between tumor cells and immune components after radiotherapy. It heralds a new era in which metabolic imaging transcends mere structural assessment, evolving into a dynamic biomarker of immune competence within the tumor microenvironment.
In summary, the upregulation of ICAM-1 following radiotherapy acts as a molecular rheostat coordinating T cell clustering via ICAM-1–LFA-1 interactions. This interaction potentiates PI3K–AKT–mTOR-driven metabolic reprogramming of T cells, boosting their glycolytic activity and tumor infiltration. The resultant surge in ^18F-FDG uptake visualized on PET scans—the metabolic flare—is thus an emblem of active antitumor immune engagement, redefining how clinicians should interpret post-RT metabolic imaging.
The implications of these findings extend beyond diagnostic imaging, inviting a reconceptualization of radiotherapy’s immunomodulatory effects and instigating the design of more rational, metabolism-aware combination therapies. Ultimately, this research bridges critical gaps in understanding the interplay between radiotherapy, immune dynamics, and tumor metabolism, spotlighting ICAM-1 as a linchpin in orchestrating effective antitumor T cell responses.
Subject of Research: Not applicable
Article Title: ICAM-1 promotes T cell glycolytic reprogramming and tumor infiltration to drive 18F-FDG PET flares following radiotherapy
News Publication Date: 19-Dec-2025
Web References: http://dx.doi.org/10.1093/procel/pwaf111
Image Credits: HIGHER EDUCATION PRESS
Keywords: Cell biology
Tags: 18F-FDG PET imaging in oncologycancer immunometabolismICAM-1 tumor microenvironmentimmune cell clustering in tumorsintracellular adhesion molecule-1 functionmetabolic reprogramming of T cellsmolecular mechanisms of radiotherapy responsePET signal surges post-radiotherapyradiotherapy and immune modulationradiotherapy-induced metabolic flareT cell metabolism in cancertumor infiltration by immune cells