In the rapidly evolving field of developmental biology, mouse embryonic stem cells (ESCs) have served as indispensable tools, primarily due to their abilities to proliferate indefinitely and differentiate into multiple cell types—a property known as pluripotency. However, despite these remarkable features, ESCs are limited in their potential to generate extraembryonic tissues such as the placenta and yolk sac, critical components that support embryonic development in vivo. This limitation has spurred intensive research aimed at pushing the boundaries of cellular potency toward totipotency—the highest developmental potential a cell can possess—which encompasses the ability to form both embryonic and extraembryonic lineages. Recent groundbreaking work has shed light on the pivotal role of the gene Hmgn3 in bestowing mouse ESCs with totipotency-like characteristics, thereby opening new avenues for studies in early embryogenesis and synthetic embryology.
Totipotency, a rare and transient state during the earliest stages of embryonic development, is characterized by the capacity to develop into all cell types that constitute the organism as well as the supportive extraembryonic structures necessary for survival and growth. This state is distinct from pluripotency, which allows cells to give rise to all embryonic tissues but excludes extraembryonic derivatives. Achieving stable totipotency or totipotent-like states in vitro has proven challenging, yet vital for advancing regenerative medicine, understanding developmental biology, and engineering artificial embryos with full developmental potential. Previous strategies to induce totipotent-like states have predominantly centered around optimizing culture conditions, but the molecular mechanisms governing this process remain inadequately understood.
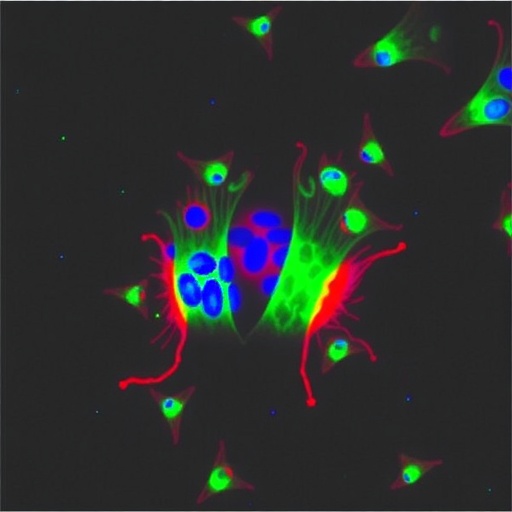
In a recent pivotal study, Wang and colleagues successfully engineered a mouse embryonic stem cell line with forced overexpression of the gene Hmgn3 (Hmgn3-OE ESCs). This line exhibited enhanced plasticity and developmental versatility compared to its wild-type counterparts. Importantly, these modified ESCs, when incorporated into developing embryos in chimera assays, demonstrated stable and robust contributions not only to fetal tissues but also to essential extraembryonic structures including the placenta and yolk sac. This finding represents a significant leap in stem cell biology, underscoring the capacity of a single gene, Hmgn3, to activate a totipotency-like program in ESCs.
To further characterize the totipotent features of Hmgn3-overexpressing ESCs, the researchers explored their behavior in vitro by inducing the formation of blastoid-like structures, or blastoids. These three-dimensional structures recapitulate critical aspects of natural blastocyst architecture and function. Notably, Hmgn3-OE ESCs self-organized into blastoids that closely resembled wild-type blastocysts at the cellular and molecular levels. The blastoids exhibited appropriate lineage segregation and expression profiles akin to genuine embryos, thereby validating the totipotency and developmental competence of the engineered ESCs. Such artificial embryo models provide a powerful platform for dissecting early developmental events and evaluating gene function under controlled conditions.
One of the most compelling discoveries of this study lies in the elucidation of the downstream regulatory network orchestrated by Hmgn3. The team identified the gene Dux, known as a key driver of totipotency-associated gene expression, as a critical mediator of Hmgn3’s effect. Loss-of-function experiments revealed that knockout of Dux substantially diminished the enhanced totipotency phenotype of Hmgn3-OE ESCs, highlighting the gene’s indispensable role in the molecular cascade activated by Hmgn3. This interplay between Hmgn3 and Dux underscores a tightly regulated genetic axis that governs the acquisition and maintenance of totipotent states.
From a mechanistic standpoint, Hmgn3 likely functions as an important epigenetic modulator influencing chromatin architecture and transcriptional accessibility. Its overexpression could facilitate the opening of chromatin domains associated with totipotency-related genes, thereby enabling ESCs to activate gene expression programs typical of totipotent cells. This chromatin remodeling potentially underpins the observed phenotypic plasticity and enhanced developmental potential. Understanding these epigenetic modifications offers profound insights into the control of cell fate decisions and regulatory networks in early embryogenesis.
Moreover, this research provides compelling evidence supporting the feasibility of reconstructing embryonic development programs using engineered stem cells. The ability of Hmgn3-OE ESCs to form both fetal and extraembryonic structures highlights their potential utility in modeling complex developmental processes in vitro, surpassing the capabilities of conventional ESCs. Such models may pave the way for novel experimental approaches to study genetic diseases, embryonic patterning, and lineage specification without the ethical concerns associated with using actual human embryos.
Importantly, the implications of activating totipotency in stem cells extend beyond fundamental biology into practical biomedical applications. Totipotent-like stem cells could revolutionize regenerative medicine by offering comprehensive tools for tissue engineering, disease modeling, and cell-based therapies that require the generation of diverse cell types and supportive tissues. Additionally, these advances could expedite the development of synthetic embryos as testing platforms for drug discovery and toxicology.
Looking forward, further research is needed to explore the precise molecular mechanisms by which Hmgn3 modulates chromatin and transcription, as well as the interplay with other critical factors in the regulation of totipotency. Additionally, expanding these findings to human stem cells could have transformative impacts on developmental biology and regenerative therapies. The integration of genome editing technologies with the insights from this study may provide unprecedented control over stem cell potency and developmental trajectories.
In summary, the pioneering work by Wang et al. marks a paradigm shift in stem cell biology, demonstrating that a single gene, Hmgn3, can significantly elevate the developmental capacity of mouse embryonic stem cells to a totipotent-like state. By delineating the molecular circuit involving Hmgn3 and its downstream effector Dux, this research lays a robust foundation for engineering artificial embryo models and advancing our understanding of the earliest stages of life. The ability to reliably generate totipotent-like cells in vitro heralds a new era in developmental and regenerative medicine, with implications that are as profound as they are promising.
Subject of Research: Induction of totipotency in mouse embryonic stem cells via Hmgn3 overexpression
Article Title: Hmgn3 is critical for inducing totipotency in mouse embryonic stem cells
Web References:
DOI: 10.1016/j.scib.2025.10.025
Image Credits: ©Science China Press
Keywords: totipotency, embryonic stem cells, Hmgn3, Dux, blastoid, extraembryonic tissues, developmental biology, chromatin remodeling, artificial embryo models, mouse ESCs
Tags: cellular potency enhancementdevelopmental biology breakthroughsembryogenesis research advancementsembryonic and extraembryonic lineagesextraembryonic tissue developmentHmgn3 gene functionin vitro totipotency inductionmouse embryonic stem cellspluripotency versus totipotencystem cell differentiation potentialsynthetic embryology implicationstotipotency in stem cells