Paving way for new preventive measures for tooth decay
Credit: HKUST
An inter-disciplinary team of researchers led by Prof. Qian Peiyuan, Chair Professor at the Hong Kong University of Science and Technology (HKUST)’s Department of Ocean Science and Division of Life Science has unraveled how a novel microbial small molecule released by Streptococcus mutans (S. mutans) – a bacterium commonly found in the human oral cavity – is connected to dental caries development using a synthetic biology approach, offering new insights to the health impact of the human oral microbiota and facilitating future research on the prevention of tooth decay. The research findings were recently published in the prestigious scientific journal Nature Chemical Biology and reported by Nature as one of the research highlights.
Every wetted surface on our planet is covered by biofilm made of microbial cells meshed in extracellular organic matrix. An early study by the National Institutes of Health (NIH) concluded that over 80% of human bacterial infection were caused by biofilm . Thus, S. mutans, a natural and primary inhabitant of the human oral cavity that has strong ability to form biofilms and produce organic acids, has long been acknowledged as the major etiological agent of dental caries.
The development of dental caries, or tooth decay, is a complex process that mainly depends on the presence of microbial biofilms on the tooth surfaces, which are known as dental plaque. Tooth decay has been recognized as one of the most common bacterial infections and costly chronic conditions afflicting humans. Annually, the global economic burden of treating tooth decay amounts to billions of dollars(1) . Although the macromolecular agents of S. mutans for biofilm formation and development have been extensively investigated, the role of small-molecule secondary metabolites in biofilm formation of S. mutans remains largely unexplored.
Prof. Qian’s research team has been studying the microbe-animal interactions mediated by signal molecules from biofilm, using integrated genomics, transcriptomics and chemical biology approaches. Recently, the research team has extended their work on biofilms related to public health.
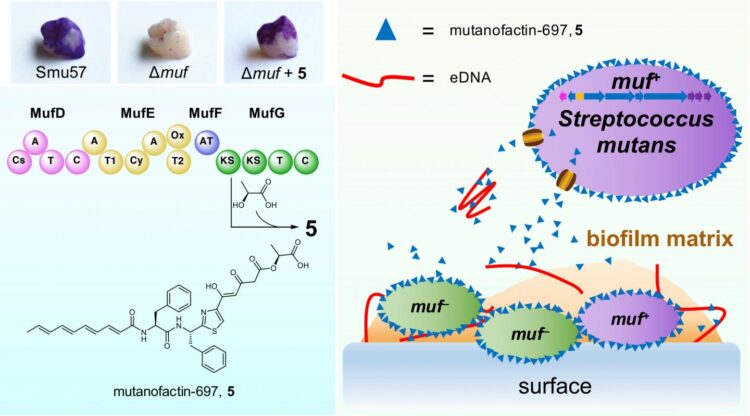
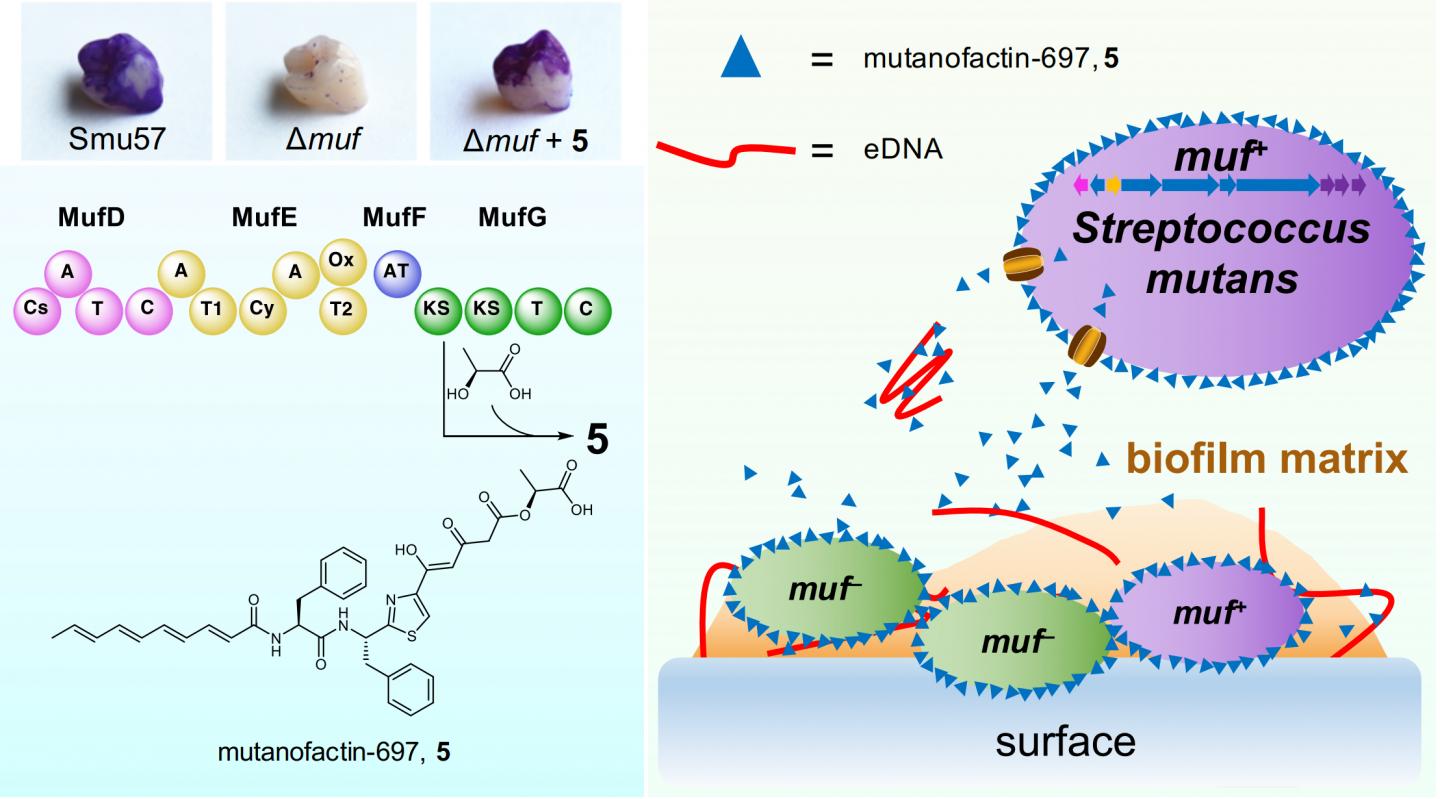
In collaboration with Prof. ZHANG Wen-Jun and Prof. Roya MABOUDIAN at University of California at Berkeley, and Prof. Robert BURNE at College of Dentistry, University of Florida, the team has discovered a polyketide/non-ribosomal peptide biosynthetic gene cluster, muf, which directly correlates with a strong biofilm-forming capability, from S. mutans strains clinically isolated from dental plaque. Then, the muf-associated bioactive product, mutanofactin-697 that contains a novel molecular scaffold was identified. Further mode-of-action studies revealed that this unique microbial secondary metabolite promotes biofilm formation via an unprecedented physicochemical mechanism: this small molecule binds to S. mutans cells and extracellular DNA, increases bacterial hydrophobicity, and subsequently promotes bacterial adhesion and biofilm formation.
Prof. Qian, also David von Hansemann Professor of Science at HKUST, said, “Our findings provide the first example of a microbial secondary metabolite promoting biofilm formation via a physicochemical approach, highlighting the significance of secondary metabolism in mediating critical processes related to the development of dental caries.”
LI Zhongrui, a researcher of the team, said this discovery will enable further mechanistic exploration of mutanofactin-related chemical regulatory processes in human oral ecology and streptococci-induced dental caries incidence and prevention.
###
(1) Global oral health burden amounts to $442 billion
https:/
Media Contact
Lindy Wong
[email protected]
Original Source
http://hkust.
Related Journal Article
http://dx.