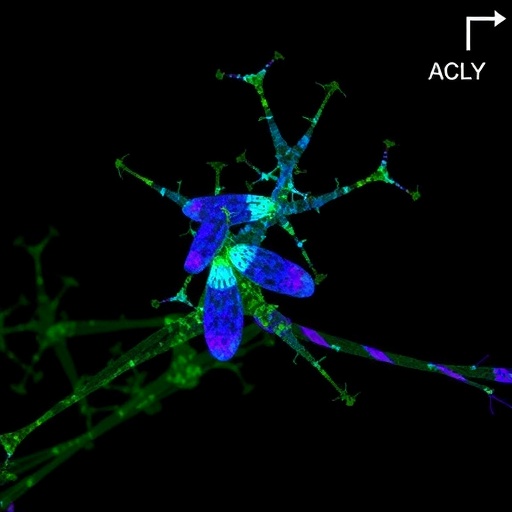
In a groundbreaking study that could redefine our understanding of brain cell development, researchers have unveiled a compelling link between glucose metabolism and the intricate dance of oligodendrocyte progenitor cell (OPC) fate decisions. This discovery provides fresh insights into how these vital progenitor cells decide whether to survive, multiply, or mature into myelin-forming oligodendrocytes, a process essential for proper neural function and repair. The research, published in the prestigious journal Nature Neuroscience, highlights the role of metabolic activity—specifically fluctuating glucose levels—in regulating OPC dynamics through enzyme ACLY-mediated histone acetylation.
For decades, neuroscientists grappled with the mystery of how OPCs orchestrate their population dynamics during brain development and repair. OPCs are the precursors to oligodendrocytes, the specialized cells responsible for forming myelin sheaths that insulate nerve fibers, enhancing electrical signal transmission. However, the cues guiding OPCs to either remain as progenitors, proliferate, or differentiate into oligodendrocytes were largely elusive. This study pioneers the concept that metabolic environment, shaped by glucose availability and vascular changes, profoundly influences OPC behavior and epigenetic regulation.
The investigators meticulously charted glucose concentrations across various brain regions and developmental stages, revealing striking spatial and temporal variations. Regions bathed in high glucose levels showcased robust OPC proliferation, accompanied by heightened histone acetylation—a hallmark of active gene transcription—suggesting a direct metabolic influence on gene expression regulation. Conversely, areas with lower glucose exhibited reduced OPC proliferation and histone acetylation, underscoring the critical role of glucose as a molecular modulator in the neural landscape.
At the molecular crossroads of this regulation lies ATP-citrate lyase (ACLY), an enzyme serving as a metabolic gatekeeper. ACLY converts citrate derived from glucose metabolism into acetyl-CoA, a central metabolic intermediate that fuels histone acetylation within the nucleus. The study’s authors demonstrated that in OPCs, ACLY-generated nuclear acetyl-CoA is indispensable for promoting histone acetylation, thereby facilitating gene expression programs that drive OPC proliferation. This enzymatic activity ties cellular metabolic state directly to chromatin remodeling and developmental gene regulation, forging a novel epigenetic-metabolic nexus.
To probe the essentiality of ACLY, the research team engineered mouse models with targeted deletion of the Acly gene specifically in OPCs. These genetically modified mice exhibited a transient hypomyelination phenotype, marked by fewer OPCs and diminished myelin formation during critical developmental windows. This phenotype elegantly corroborates that ACLY-mediated metabolic pathways are fundamental for maintaining OPC numbers through promoting their proliferation, ultimately ensuring adequate oligodendrocyte numbers for myelination.
Interestingly, despite the initial deficiency in OPCs and delayed myelination, the Acly-deficient mice showed that oligodendrocyte differentiation was not entirely halted. The study unmasked a compensatory metabolic switch wherein oligodendrocytes adopt alternative enzymatic routes for generating extranuclear acetyl-CoA from non-glucose substrates. This metabolic plasticity enables differentiation and myelin synthesis to proceed, albeit through pathways independent of ACLY. These findings underscore a remarkable adaptability of oligodendrocyte lineage cells, employing distinct metabolic routes to meet their stage-specific requirements.
This nuanced metabolic choreography reveals that OPC proliferation relies strictly on ACLY-dependent nuclear acetyl-CoA derived from glucose metabolism, distinctly linking it to epigenetic histone acetylation that governs gene expression. In contrast, mature oligodendrocytes repurpose other metabolic pathways to supply acetyl-CoA extraneously, supporting myelin biosynthesis. Such metabolic compartmentalization during the lineage progression emphasizes how cellular metabolism is intricately integrated with developmental programs and cellular differentiation.
Beyond the fundamental biology of the oligodendrocyte lineage, these insights may have profound clinical implications. Given that myelin integrity is compromised in numerous neurological disorders such as multiple sclerosis, understanding the metabolic control of OPC proliferation and differentiation could unlock new therapeutic avenues. Modulating glucose availability or targeting ACLY could potentially enhance remyelination or support repair mechanisms in demyelinating diseases.
Moreover, the discovery that vascularization influences local glucose levels adds another dimension to brain development and repair. Vascular remodeling, by altering glucose supply, directly orchestrates OPC dynamics and epigenetic states, revealing the vascular niche as a critical modulator of neural progenitor fate. This intersection of metabolism, epigenetics, and microenvironmental factors expands our appreciation of stem cell niches beyond mere biochemical signals to encompass metabolic landscapes.
The reported findings also raise intriguing questions about how metabolic states might intersect with aging or disease states in the brain. Fluctuations in glucose metabolism or vascular health during aging could perturb OPC proliferation or differentiation, thereby impacting myelin maintenance or regeneration. Future studies may explore whether metabolic interventions can rejuvenate aging OPC populations or mitigate neurodegenerative processes via epigenetic reprogramming.
Technically, the study employed a sophisticated combination of metabolic profiling, epigenetic assays, and genetic manipulations, producing a comprehensive picture of how fluctuating glucose levels are translated into epigenetic marks that govern cellular outcomes. The identification of ACLY as a key enzymatic hub further provides a tangible molecular target for potential manipulation. This integrative approach exemplifies the power of combining cell biology, epigenetics, metabolism, and genetics to unravel complex neurodevelopmental processes.
In summary, this groundbreaking research provides compelling evidence that glucose metabolism, through ACLY-mediated nuclear acetyl-CoA production, exerts spatially and temporally precise control over OPC proliferation via histone acetylation. The distinction between metabolic requirements for progenitor expansion versus mature oligodendrocyte function underscores a sophisticated, stage-specific metabolic-epigenetic regulation in the oligodendrocyte lineage. These discoveries pave the way for novel metabolic and epigenetic strategies to enhance myelin repair and combat neurological diseases.
As neuroscience increasingly recognizes metabolism as a fundamental regulator of cell fate and function, this study situates OPC biology within an exciting new metabolic frontier. The explicit link between glucose, vascular modulation, ACLY function, and histone acetylation illuminates a previously underappreciated axis of developmental control. These insights hold transformative potential not only for understanding myelination but also for broader applications in cell differentiation and regeneration across the nervous system.
Future directions building on this research are poised to explore the therapeutic potential of manipulating ACLY activity or glucose metabolism in disease contexts. Investigations into additional metabolic enzymes and epigenetic modifications involved in OPC and oligodendrocyte biology could further unravel the complex networks underlying brain development. As this field advances, integrating metabolic state with genetic and epigenetic regulation will become an essential paradigm in neurobiology.
In conclusion, the elegant study by Sauma and colleagues represents a seminal step toward deciphering the metabolic code that governs OPC population dynamics and myelination. By linking glucose-dependent ACLY activity to histone acetylation and cell proliferation, it not only deepens our understanding of brain development but also unveils promising avenues for therapeutic innovation in demyelinating disorders. This metabolic lens on cell fate offers a thrilling new chapter in the quest to understand and harness the brain’s regenerative capacity.
Subject of Research:
Oligodendrocyte progenitor cell proliferation, differentiation, and metabolic regulation via glucose availability and ACLY-mediated histone acetylation.
Article Title:
Glucose-dependent spatial and temporal modulation of oligodendrocyte progenitor cell proliferation via ACLY-regulated histone acetylation.
Article References:
Sauma, S., Stransky, S., Selcen, I. et al. Glucose-dependent spatial and temporal modulation of oligodendrocyte progenitor cell proliferation via ACLY-regulated histone acetylation. Nat Neurosci (2026). https://doi.org/10.1038/s41593-026-02263-7
Image Credits:
AI Generated
DOI:
https://doi.org/10.1038/s41593-026-02263-7
Tags: ACLY enzyme role in brain developmentepigenetic control of brain repairglucose influence on neural cell fateglucose levels and OPC survivalglucose metabolism in oligodendrocyte progenitor cellshistone acetylation and OPC differentiationmetabolic regulation of myelin formationNature Neuroscience oligodendrocyte studyneural metabolism and myelinationoligodendrocyte progenitor cell proliferationOPC dynamics in brain developmentvascular impact on progenitor cell growth