“[…] the results of this preclinical investigation program suggest that induction of apoptosis is a key component of GFH009’s anti-tumor mechanism of action […]”
Credit: 2023 Zhou et al.
“[…] the results of this preclinical investigation program suggest that induction of apoptosis is a key component of GFH009’s anti-tumor mechanism of action […]”
BUFFALO, NY- December 28, 2023 – A new research paper was published in Oncotarget’s Volume 14 on December 20, 2023, entitled, “The pharmacodynamic and mechanistic foundation for the antineoplastic effects of GFH009, a potent and highly selective CDK9 inhibitor for the treatment of hematologic malignancies.”
To evade cell cycle controls, malignant cells rely upon rapid expression of select proteins to mitigate pro-apoptotic signals resulting from damage caused by both cancer treatments and unchecked over-proliferation. Cyclin-dependent kinase 9 (CDK9)-dependent signaling induces transcription of downstream oncogenes promoting tumor growth, especially in hyperproliferative ‘oncogene-addicted’ cancers, such as human hematological malignancies (HHMs). In this new study, researchers Fusheng Zhou, Lili Tang, Siyuan Le, Mei Ge, Dragan Cicic, Fubo Xie, Jinmin Ren, Jiong Lan, and Qiang Lu from GenFleet Therapeutics Inc. and Sellas Life Sciences Group aimed to summarize current knowledge underlying the mechanism of action (MOA) of GFH009 and explain its robust anti-cancer activity.
“Understanding GFH009’s MOA allows for a more optimal clinical development path, given the potential for meaningful benefits in patients with hematological malignancies.”
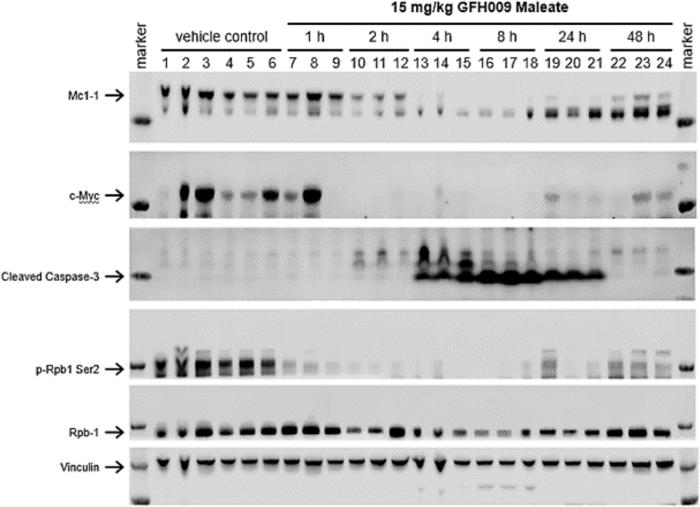
GFH009, a potent, highly selective CDK9 small molecule inhibitor, demonstrated antiproliferative activity in assorted HHM-derived cell lines, inducing apoptosis at IC50 values below 0.2 μM in 7/10 lines tested. GFH009 inhibited tumor growth at all doses compared to controls and induced apoptosis in a dose-dependent manner.
Twice-weekly injections of GFH009 maleate at 10 mg/kg significantly prolonged the survival of MV-4-11 xenograft-bearing rodents, while their body weight remained stable. There was marked reduction of MCL-1 and c-MYC protein expression post-drug exposure both in vitro and in vivo. Through rapid ‘on-off’ CDK9 inhibition, GFH009 exerts a proapoptotic effect on HHM preclinical models triggered by dynamic deprivation of crucial cell survival signals.
“Our results mechanistically establish CDK9 as a targetable vulnerability in assorted HHMs and, along with the previously shown superior class kinome selectivity of GFH009 vs other CDK9 inhibitors, strongly support the rationale for currently ongoing clinical studies with this agent in acute myeloid leukemia and other HHMs.”
Read the full paper: DOI: https://doi.org/10.18632/oncotarget.28543
Correspondence to: Jiong Lan
Email: [email protected]
Keywords: GFH009, CDK9, cancer, leukemia, cell cycle
About Oncotarget: Oncotarget (a primarily oncology-focused, peer-reviewed, open access journal) aims to maximize research impact through insightful peer-review; eliminate borders between specialties by linking different fields of oncology, cancer research and biomedical sciences; and foster application of basic and clinical science.
To learn more about Oncotarget, visit Oncotarget.com and connect with us on social media:
- X, formerly known as Twitter
- YouTube
- LabTube
- Soundcloud
Sign up for free Altmetric alerts about this article: https://oncotarget.altmetric.com/details/email_updates?id=10.18632%2Foncotarget.28543
Click here to subscribe to Oncotarget publication updates.
For media inquiries, please contact: [email protected].
Oncotarget Journal Office
6666 East Quaker Str., Suite 1A
Orchard Park, NY 14127
Phone: 1-800-922-0957 (option 2)
###
Journal
Oncotarget
DOI
10.18632/oncotarget.28543
Method of Research
Observational study
Subject of Research
Cells
Article Title
The pharmacodynamic and mechanistic foundation for the antineoplastic effects of GFH009, a potent and highly selective CDK9 inhibitor for the treatment of hematologic malignancies
Article Publication Date
20-Dec-2023