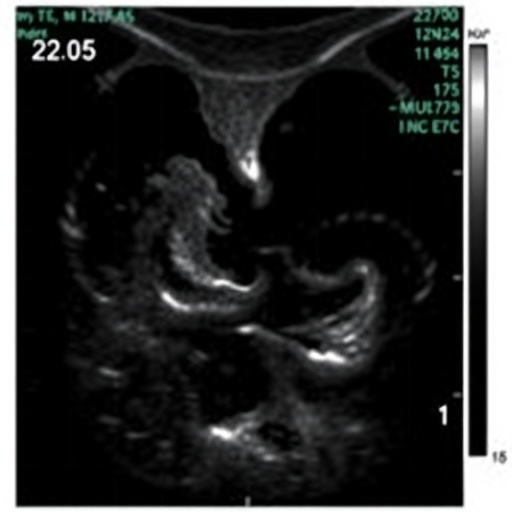
In a groundbreaking advancement within neonatal medicine, researchers have unveiled critical echocardiographic factors that can predict adverse outcomes in infants diagnosed with congenital diaphragmatic hernia (CDH). This developmental anomaly, characterized by an incomplete formation of the diaphragm, allows abdominal contents to migrate into the chest cavity, severely compromising pulmonary and cardiac function in newborns. The recently published study, led by Ting, J.Y., Sehgal, A., and Kuan, M.T.Y., dives deep into the cardiac imaging signatures that forecast poor prognosis in this vulnerable population, offering neonatologists a potent tool for early intervention and personalized therapeutic approaches.
Congenital diaphragmatic hernia represents a complex clinical challenge, as its pathophysiology hinges not only on structural lung hypoplasia but also on pulmonary hypertension and cardiac dysfunction. Historically, the emphasis has been on lung development and ventilatory strategies, yet cardiovascular complications notably influence survival and morbidity. The research team harnessed advanced echocardiographic techniques to delineate the patterns in cardiac function that correspond to mortality and morbidities such as persistent pulmonary hypertension of the newborn and long-term neurodevelopmental impairments.
Utilizing high-resolution, prospective echocardiographic assessment, the study enrolled a cohort of infants diagnosed prenatally or shortly post-birth with CDH. Serial cardiac evaluations were meticulously performed in the immediate neonatal period, focusing on parameters such as right ventricular systolic pressure, tricuspid annular plane systolic excursion (TAPSE), and left ventricular ejection fraction. These metrics offer quantitative insights into the hemodynamic burden imposed by pulmonary vascular resistance and ventricular interdependence, both of which are pivotal in modulating clinical outcomes.
One of the most salient findings highlighted by the investigators was the prognostic significance of right ventricular function markers. Elevated right ventricular systolic pressure identified via echocardiography consistently correlated with increased incidence of extracorporeal membrane oxygenation (ECMO) requirement and mortality. This finding underscores the hypothesis that right ventricular afterload, a consequence of pulmonary hypertension, strains the cardiovascular system and underlies the cascade toward clinical deterioration in infants with CDH.
Moreover, the assessment of the interventricular septal configuration revealed critical insights into ventricular interaction. Altered septal geometry during systole and diastole suggested ventricular interdependency disruption, which clearly predicted hemodynamic instability. This mechanistic understanding advances neonatal care by pinpointing which patients are at escalated risk for cardiovascular collapse and may benefit from early pharmacological or surgical interventions targeting pulmonary vascular resistance.
Additional echocardiographic indicators, such as diminished TAPSE values, also emerged as predictors of adverse short-term outcomes. TAPSE, a measure of longitudinal right ventricular function, appeared instrumental in detecting subclinical ventricular dysfunction before overt heart failure manifestations. The use of this non-invasive parameter positions echocardiography as not only diagnostic but also prognostic, aligning with precision medicine paradigms in neonatal care.
These findings propel the role of echocardiography beyond traditional diagnostic domains into a pivotal prognostic realm. The study emphasizes the necessity of integrating comprehensive cardiac evaluation into the multidisciplinary management strategy for infants with CDH. As a result, cardiologists and neonatologists can jointly refine risk stratification models, tailor respiratory and hemodynamic support modalities, and monitor therapeutic efficacy dynamically.
Further elucidation through multivariate analysis confirmed that echocardiographic variables operated independently of conventional risk markers such as birth weight, gestational age, or lung-to-head ratio measured prenatally. This dissociation identifies cardiac function as an autonomous determinant of clinical trajectory, advocating for routine cardiac imaging as part of standardized postnatal assessments.
The implications of this study extend into shaping future clinical trials and therapeutic guidelines. Targeted therapies addressing right ventricular afterload reduction and functional support can now be more precisely timed, potentially enhancing survival rates and reducing long-term sequela in infants with CDH. Precision in patient selection for interventions like ECMO could also be refined, mitigating procedural risks and optimizing resource allocation.
Notably, the authors highlighted the potential for novel echocardiographic technologies, including speckle-tracking and three-dimensional imaging, to further enhance the characterization of cardiac mechanics in CDH. Such advanced modalities could augment the predictive accuracy and offer more nuanced insights into myocardial deformation patterns, expanding the clinical utility of bedside cardiac ultrasound.
Despite the promise, the authors caution that echocardiographic assessments require expertise and standardized protocols to ensure reproducibility and accuracy. Training initiatives and consensus guidelines may be necessary to embed these evaluation strategies into routine neonatal intensive care units globally, facilitating widespread adoption and impact.
In conclusion, this pivotal research underscores echocardiographic evaluation as an indispensable frontier in newborn care for congenital diaphragmatic hernia. By illuminating cardiac functional hallmarks that presage adverse clinical outcomes, the study redefines risk assessment and therapeutic stratagems, with the ultimate goal of improving survival and quality of life for affected infants. The integration of cardiovascular insights will indubitably enrich multidisciplinary management approaches and inspire further innovation in neonatal medicine.
As clinicians and researchers digest these findings, a new paradigm is emerging—one that acknowledges the heart’s centrality in the pathogenesis and prognosis of CDH. This evolution in understanding portends a future where precision diagnostics and targeted cardiac therapeutics transform neonatal outcomes, making what was once a dire diagnosis increasingly manageable and survivable.
Subject of Research: Echocardiographic predictors of adverse outcomes in infants with congenital diaphragmatic hernia
Article Title: Echocardiographic predictors of adverse outcomes in infants with congenital diaphragmatic hernia
Article References:
Ting, J.Y., Sehgal, A., Kuan, M.T.Y. et al. Echocardiographic predictors of adverse outcomes in infants with congenital diaphragmatic hernia. J Perinatol (2026). https://doi.org/10.1038/s41372-026-02670-5
Image Credits: AI Generated
DOI: 14 April 2026
Tags: advanced neonatal echocardiography techniquescardiac dysfunction in congenital diaphragmatic herniacongenital diaphragmatic hernia echocardiographic predictorsechocardiographic markers for infant prognosisechocardiography for neonatal risk assessmentneonatal cardiac imaging in CDHneonatal pulmonary hypoplasia evaluationneurodevelopmental outcomes in CDH infantspersistent pulmonary hypertension of the newbornpersonalized therapy for neonatal CDHprenatal diagnosis of CDHpulmonary hypertension in newborns