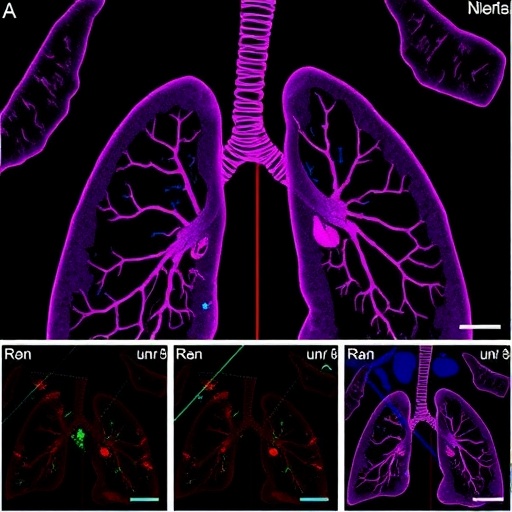
In a groundbreaking development poised to transform the therapeutic landscape for idiopathic pulmonary fibrosis (IPF), researchers at Virginia Tech’s Fralin Biomedical Research Institute have unveiled a novel strategy that not only halts but also reverses the progression of lung scarring. Published recently in the prestigious journal Theranostics, the study unveils the critical role of two proteins, ID1 and ID3, whose simultaneous inhibition demonstrates remarkable efficacy in mitigating fibrosis—a condition notorious for its relentless deterioration of lung function.
Idiopathic pulmonary fibrosis is a devastating disease characterized by progressive scarring of the lung tissue, leading to severe respiratory impairment and a dismal prognosis. Current treatments manage to slow the disease but fall short of reversing established fibrosis, leaving patients with a median survival of three to five years post-diagnosis. The Virginia Tech team’s approach signifies a pivotal shift, targeting the molecular underpinnings of fibrosis at the cellular level, promising a new horizon in pulmonary medicine.
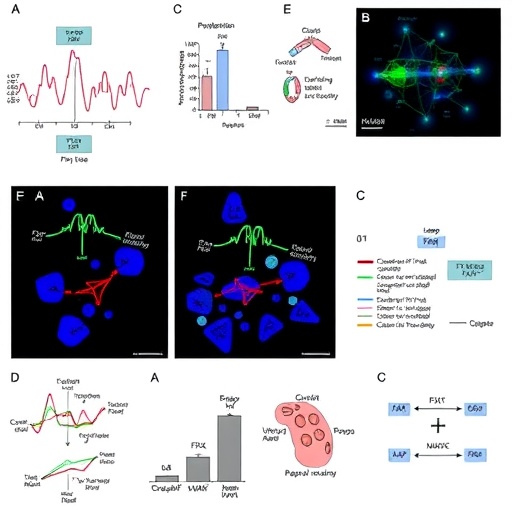
The study, spearheaded by assistant professor Yassine Sassi, integrates comprehensive analyses of human lung tissues and fibroblast cells derived from IPF patients with extensive preclinical models in mice. Their findings reveal heightened expression of ID1 and ID3 in diseased lungs, identifying these proteins as pivotal drivers of fibroblast activation—a central event in fibrotic tissue remodeling. The manipulation of these targets was achieved through both small-molecule inhibitors and innovative gene therapy techniques, offering versatile avenues for clinical translation.
Notably, fibroblasts, the architects of scar tissue in IPF, exhibit pathological hyperactivation mediated by signaling cascades involving the MEK/ERK pathway and alterations in cell cycle dynamics. ID1 and ID3 function as regulators within these processes, orchestrating fibroblast proliferation and extracellular matrix deposition. By concurrently suppressing these proteins, the research team effectively dismantled the feedback loops perpetuating fibrotic progression, thereby restoring tissue homeostasis.
The therapeutic interventions were tested rigorously across multiple experimental platforms. Remarkably, both pharmacological and genetic modalities yielded significant attenuation of lung fibrosis in murine models, with some outcomes paralleling or surpassing those achieved by FDA-approved antifibrotic agents. These findings underscore the potential for ID1 and ID3 inhibitory strategies to not only halt fibrotic augmentation but also to induce regression of pre-existing lesions, a feat unprecedented in current clinical practice.
Mechanistically, the dual inhibition impedes aberrant fibroblast cycling by targeting pathways essential for cell division and survival, including the MEK/ERK axis known for its implication in fibrotic and carcinogenic processes. This focused disruption halts the relentless expansion and activation of fibroblast populations, thereby curtailing scar matrix accumulation. Such intricately targeted approaches highlight a paradigm shift from broad-spectrum antifibrotic agents toward precision medicine.
The research extends beyond bench-based findings; it incorporates collaborative expertise spanning institutions such as the Icahn School of Medicine at Mount Sinai and Boston University, amplifying its translational relevance. The multidisciplinary collaboration ensured that insights derived from patient-derived samples were seamlessly integrated with sophisticated animal models, enriching the robustness of the conclusions.
Importantly, the study elucidates a fundamental aspect of pulmonary fibrosis pathophysiology—how intracellular interactions involving ID1 and ID3 fuel the disease’s relentless course. This molecular insight brings clarity to previously obscure mechanisms, enabling the design of next-generation therapeutics that can intervene more effectively in the disease cascade.
Despite the monumental progress reported, researchers are mindful of the complexity inherent in translating these findings into clinical therapies. Challenges remain regarding drug delivery, off-target effects, and the long-term safety profile of ID1 and ID3 inhibitors. Nonetheless, the study lays a strong conceptual and experimental foundation that will guide future clinical trials aimed at delivering transformative benefits to patients.
The implications of this research reverberate beyond IPF. Fibrotic processes, propelled by similar pathogenic pathways, are central to a spectrum of chronic conditions affecting vital organs such as the liver, heart, and kidneys. Thus, the identification of ID1 and ID3 as fibrosis drivers opens avenues for broad therapeutic innovation, potentially impacting a myriad of fibrotic diseases.
In conclusion, the Virginia Tech team’s pioneering work, led by Yassine Sassi and carried out with the dedication of postdoctoral fellow Samar Antar and research associate Jacob Dahlka, represents a milestone in respiratory medicine. By targeting ID1 and ID3, they have illuminated a new path toward reversing lung fibrosis, offering hope to thousands afflicted by this intractable condition. Their work emboldens the scientific community’s pursuit of targeted, effective, and reversible interventions for pulmonary fibrosis and beyond.
Subject of Research: People
Article Title: Simultaneous inhibition of ID1 and ID3 mitigates fibroblast activation via cell cycle and MEK/ERK pathways in pulmonary fibrosis
News Publication Date: 16-Apr-2026
Web References:
Theranostics Journal Article
DOI: 10.7150/thno.127118
Image Credits: Clayton Metz/Virginia Tech
Keywords: Idiopathic pulmonary fibrosis, lung fibrosis, ID1 and ID3 inhibitors, fibroblast activation, MEK/ERK pathway, cell cycle regulation, therapeutic development, targeted gene therapy, small-molecule inhibitors, fibrosis reversal, pulmonary medicine, fibrotic diseases
Tags: dual protein inhibition in lung fibrosisfibroblast activation in lung diseaseID1 and ID3 protein role in fibrosisidiopathic pulmonary fibrosis treatmentlung fibrosis drug developmentnovel therapies for lung scarringpreclinical models of IPFpulmonary fibrosis molecular targetsreversing lung scarring preclinical studyTheranostics journal fibrosis studytherapeutic strategies for pulmonary fibrosisVirginia Tech lung fibrosis research