Scientists at the renowned Wistar Institute have pioneered an innovative approach to enhance the efficacy of cancer treatments by engineering a novel small molecule drug conjugate capable of selectively targeting tumors with higher precision. At the heart of this breakthrough lies the conjugation of an Aurora kinase A (AURKA) inhibitor, a molecule known for its ability to arrest tumor growth by disrupting cell division, with a tumor-targeting moiety that binds to heat shock protein 90 (HSP90), a protein abundantly expressed in cancer cells. This strategic combination aims to increase drug concentration within tumoral tissue while minimizing adverse effects on healthy cells—a longstanding challenge in oncology therapeutics.
Aurora kinase A plays a pivotal role in the regulation of mitotic events essential for cell proliferation, making it a prime target for cancer intervention. However, clinical application of AURKA inhibitors has been disproportionately hampered by systemic toxicity, as the inhibitors do not sufficiently discriminate between malignant and non-malignant tissues. Recognizing these limitations, the Wistar Institute team, led by Dr. Joseph Salvino, conceptualized a molecular ‘Lego’ strategy, where the AURKA inhibitor was chemically linked to an HSP90-binding molecule to forge a chimeric compound dubbed NN-01-195. This design exploits the overexpression of HSP90 in tumors to preferentially shuttle the drug to cancer cells, thereby potentially mitigating the dose-limiting toxicity observed in earlier trials.
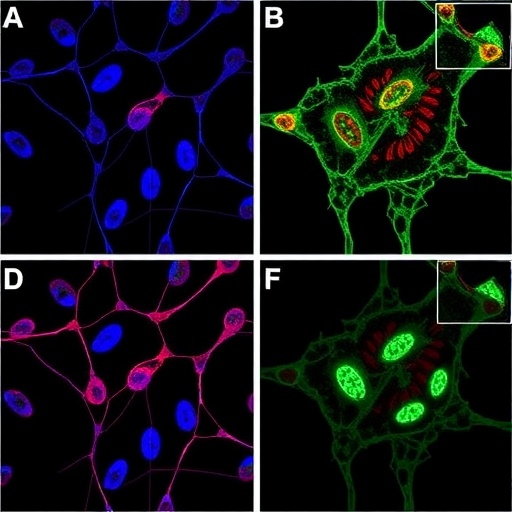
The research underpinning NN-01-195’s development involved intricate molecular engineering to achieve dual recognition of AURKA and HSP90 proteins. Rigorous in vitro analysis on diverse cancer cell lines, including those derived from head and neck squamous cell carcinoma, non-small cell lung cancer, and melanoma, demonstrated that this conjugate effectively interrupted malignant cell cycle progression. By halting critical mitotic pathways, NN-01-195 induced potent cytotoxicity confined to cancer cells, showcasing its promise as a next-generation targeted therapy.
Progressing to in vivo models, the investigational compound exhibited remarkable pharmacokinetic advantages. Quantitative studies revealed a tenfold increase in tumor accumulation of NN-01-195 compared to the unconjugated AURKA inhibitor counterpart. Furthermore, this molecule demonstrated extended tumor retention, remaining pharmacologically active 24 hours post-administration, a marked improvement over the rapid clearance profile typically seen with monotherapy AURKA inhibitors. Crucially, these preclinical evaluations identified no significant toxicities, underscoring a favorable safety profile that augurs well for subsequent clinical translation.
Another compelling facet of this investigation was the observed synergy between NN-01-195 and WEE1 kinase inhibitors, agents that disrupt cell cycle checkpoints and DNA damage repair mechanisms. When used in combination, these drugs exerted amplified suppression of tumor growth, highlighting a potential combinatorial treatment paradigm that leverages complementary molecular vulnerabilities within cancer cells. This discovery opens avenues for designing robust multi-modal regimens tailored to overcome resistance and improve patient outcomes.
Pharmacokinetics, the study of drug absorption, distribution, metabolism, and excretion, remains a critical bottleneck in drug development, with poor tumor exposure accounting for nearly half of clinical trial failures in oncology therapeutics. NN-01-195’s enhanced tumor bioavailability exemplifies how rational drug design can overcome pharmacokinetic challenges by exploiting tumor-specific markers such as HSP90. This targeted delivery not only optimizes therapeutic potency but also diminishes systemic exposure, ultimately reducing collateral damage to normal tissues.
The implications of this research extend far beyond the cancer types initially studied, given that HSP90 and AURKA are ubiquitously involved in the molecular pathology of numerous solid tumors. The modular nature of the conjugate also suggests scalability, where alternative inhibitory molecules could be tethered to tumor-targeting entities, custom-tailored to distinct oncogenic profiles. This modular platform technology thus holds transformative potential in personalized medicine, allowing therapies to be finetuned to the molecular signatures of the patient’s tumor.
Looking forward, the research team is focused on refining NN-01-195 into an orally administrable formulation, which would significantly improve patient compliance and enable chronic dosing regimens. Oral bioavailability presents a set of unique challenges including absorption stability and metabolic degradation, but success in this realm would represent a landmark advancement that could reshape the therapeutic landscape for AURKA-targeted treatments.
Collaboration between academic institutions was vital in advancing this project, including contributions from Fox Chase Cancer Center and Yale University School of Medicine, alongside The Wistar Institute. The multidisciplinary expertise combined with robust funding from institutions such as the National Institutes of Health and the Department of Defense has been instrumental in translating these scientific concepts from bench to preclinical validation.
Publication of these findings in the highly respected journal Molecular Cancer Therapeutics positions NN-01-195 as a frontrunner in the next wave of targeted oncology therapeutics. As the scientific community eagerly anticipates further clinical trials, this work underscores the promise of smartly engineered small molecule conjugates in revolutionizing cancer care, emphasizing precision, tolerability, and efficacy.
Beyond the laboratory, Wistar Institute scientists continue to push the boundaries of biomedical research, striving to tackle the most intractable challenges in cancer therapy through innovation and discovery. The advancement of NN-01-195 not only epitomizes these efforts but also provides hope for more effective and safer cancer therapies in the near future.
Subject of Research: Animals
Article Title: NN-01-195, a novel conjugate of HSP90 and AURKA inhibitors effectively targets solid tumors
News Publication Date: 23-Jan-2026
Web References:
Wistar Institute: https://www.wistar.org/
Article DOI: http://dx.doi.org/10.1158/1535-7163.MCT-25-0857
Image Credits: The Wistar Institute
Keywords: Proteins
Tags: Aurora kinase A inhibitorsCancer Treatment Innovationchimeric compounds in oncologyenhancing chemotherapy efficacyheat shock protein 90 in cancerminimizing systemic toxicity in cancer therapynovel cancer drug developmentprecision medicine for cancer treatmentsmall molecule drug conjugatestargeted drug delivery in oncologytumor-selective therapeuticsWistar Institute cancer research