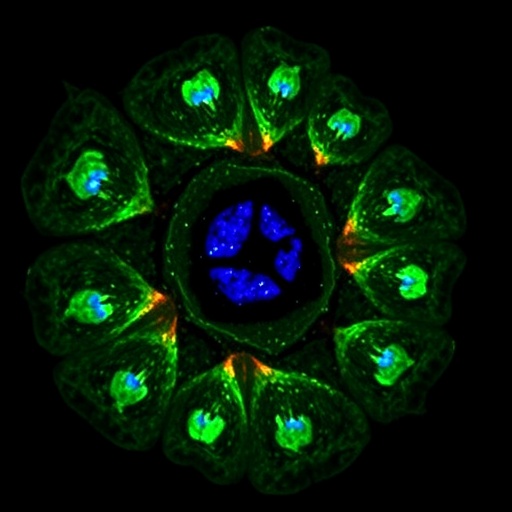
In a groundbreaking new study published in Nature Communications, researchers have unraveled a pivotal mechanism linking immune cell dysfunction to the progression of liver fibrosis, a major cause of chronic liver disease worldwide. The team, led by Al Sayegh, Wan, and Caër, among others, highlights the critical role of defective autophagy within CD4 T cells and its unexpected influence on promoting type 3 inflammation, which ultimately drives fibrotic changes in liver tissue. This discovery opens a promising frontier for targeted therapeutic strategies aimed at halting or reversing liver fibrosis by correcting immune cell autophagy defects.
Liver fibrosis is a pathological condition characterized by excessive accumulation of extracellular matrix proteins that disrupts normal liver architecture and function. It is often a progressive consequence of chronic liver injury caused by viral infections, alcohol abuse, or metabolic syndromes. Despite its global health burden, current treatments are limited, primarily focusing on managing underlying causes rather than directly intervening in the fibrotic process itself. The novel insights from this study shed light on an immune-mediated pathway that may be exploited to develop much-needed antifibrotic therapies.
The key finding centers on autophagy, a highly conserved cellular degradation process instrumental in maintaining cellular homeostasis by recycling damaged organelles and proteins. While autophagy’s role in hepatocytes and stellate cells within the liver has been extensively studied, its function in immune subsets, particularly CD4 T lymphocytes, remained elusive until now. The authors demonstrated that impaired autophagy in CD4 T cells — crucial orchestrators of adaptive immunity — triggers a pro-fibrogenic inflammatory milieu dominated by type 3 inflammation characterized by elevated interleukin-17 (IL-17) and related cytokines.
Using sophisticated genetic mouse models with targeted deletions in essential autophagy genes specifically within CD4 T cells, the researchers observed exaggerated liver fibrosis upon exposure to fibrogenic stimuli. Interestingly, this fibrotic escalation was accompanied by a marked increase in type 3 inflammatory responses, implicating a direct causative link between T cell autophagy defects and the inflammatory driver of fibrosis. This challenges prior conceptions that primarily focused on innate immune cells and hepatic stellate cell activation, repositioning CD4 T cell dysfunction as a central actor in fibrogenesis.
Further molecular analyses revealed that defective autophagy in CD4 T cells leads to the accumulation of dysfunctional mitochondria, resulting in increased mitochondrial reactive oxygen species (ROS) production. These ROS act as signaling molecules that skew T cell differentiation toward a pro-inflammatory Th17 phenotype, known for secreting IL-17. The persistent presence of IL-17 and other type 3 cytokines promotes recruitment and activation of fibroblasts and myofibroblasts in the liver, accelerating the deposition of collagen and extracellular matrix components that form fibrotic scar tissue.
Crucially, the study also examined human liver biopsy samples from patients with various stages of fibrosis and found patterns consistent with the murine data. CD4 T cells derived from fibrotic liver tissues exhibited signs of impaired autophagy and heightened type 3 inflammatory signatures. This translational aspect affirms the clinical relevance of the findings and provides a rationale for targeting autophagy pathways in CD4 T cells as a novel therapeutic intervention to mitigate liver fibrosis progression in humans.
The interplay between immune cell metabolism and function is increasingly recognized as integral to understanding chronic inflammatory diseases, and this study adds a significant chapter to that narrative. By identifying defective autophagy as a metabolic fault line that fuels pathological inflammation, the research underscores the importance of autophagic homeostasis in immune competence and tissue health. It also offers a plausible explanation for why certain individuals with chronic liver insults progress rapidly to fibrosis while others maintain relatively stable liver function.
Targeting autophagy presents unique challenges due to the pathway’s ubiquitous and complex nature. However, this work provides a focused target – CD4 T cells – where restoring autophagic flux might recalibrate immune responses and reduce fibrogenesis without broadly suppressing immunity. Pharmacological agents or genetic therapies designed to enhance autophagy selectively in T cells could balance pro- and anti-inflammatory signals, thereby halting the chronic injury cycle that drives fibrosis.
The implications of this study extend beyond liver disease, as defective autophagy within immune cells is implicated in multiple inflammatory and autoimmune conditions. By elucidating the mechanistic link between T cell autophagy dysfunction and pathological inflammation, the findings may stimulate broader investigations into how autophagy modulation can be leveraged therapeutically across diverse diseases characterized by immune dysregulation, such as multiple sclerosis, rheumatoid arthritis, and inflammatory bowel disease.
Moreover, understanding how autophagy influences T cell differentiation toward specific helper subsets provides a fundamental insight into immune cell biology. The skewing toward a Th17 phenotype upon autophagy impairment reveals how intracellular quality control machinery intersects with fate decisions that govern immunity or pathology. This concept may inspire novel strategies in vaccine development and immunotherapy where tuning T cell responses is critical for success.
In parallel with the biological discoveries, the study utilized advanced single-cell RNA sequencing and metabolic profiling, enabling the dissection of T cell populations at unprecedented resolution. These methodologies were critical in identifying the heterogeneity of T cell subsets in fibrotic livers and pinpointing metabolic defects linked to autophagy failure. Such high-dimensional analyses represent a new gold standard for immunological studies in complex diseases and facilitate the identification of biomarkers for disease staging and treatment response.
Continued research in this vein will be essential to translate these fundamental findings into clinical applications. Important next steps include designing small molecules or biologics that specifically restore autophagy in CD4 T cells without off-target effects. Additionally, clinical trials will be necessary to evaluate whether modulating autophagy ameliorates fibrosis progression or even promotes regression in patients with chronic liver diseases.
As liver fibrosis often precedes cirrhosis and liver cancer, interventions that address its immunological underpinnings hold promise for altering disease trajectories and improving patient outcomes. The work by Al Sayegh and colleagues represents a significant leap toward that goal, merging cell biology, immunology, and clinical insights to chart a new path in liver disease research.
Subject of Research: Role of defective autophagy in CD4 T cells in driving liver fibrosis via type 3 inflammation.
Article Title: Defective autophagy in CD4 T cells drives liver fibrosis via type 3 inflammation.
Article References:
Al Sayegh, R., Wan, J., Caër, C. et al. Defective autophagy in CD4 T cells drives liver fibrosis via type 3 inflammation. Nat Commun 16, 3860 (2025). https://doi.org/10.1038/s41467-025-59218-y
Image Credits: AI Generated
Tags: antifibrotic therapy developmentautophagy in immune cellsCD4 T cell dysfunctioncellular homeostasis maintenancechronic liver disease researchchronic liver injury causesextracellular matrix accumulationimmune-mediated liver injuryliver fibrosis mechanismsnovel insights in liver treatmenttargeted therapeutic strategiestype 3 inflammation and fibrosis