DURHAM, N.C. — What makes some cancer cells stay put while others break loose and spread through the body? A new study of C. elegans worms may hold some clues. Researchers at Duke University have put together what they say is the first complete “parts list” for a cell caught in the act of tunneling through the tissue barriers that normally keep cells in place.
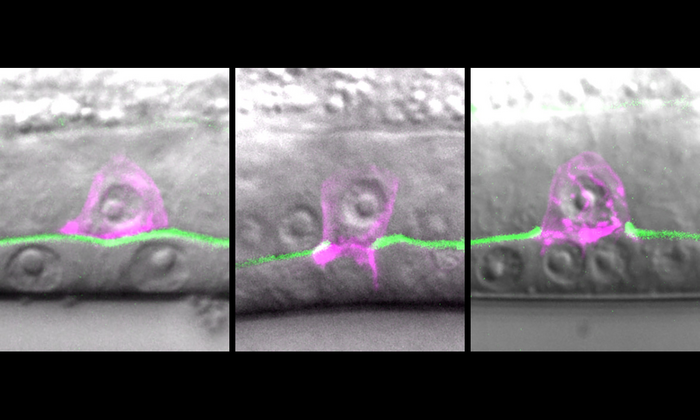
Credit: Courtesy of the Sherwood lab, Duke University
DURHAM, N.C. — What makes some cancer cells stay put while others break loose and spread through the body? A new study of C. elegans worms may hold some clues. Researchers at Duke University have put together what they say is the first complete “parts list” for a cell caught in the act of tunneling through the tissue barriers that normally keep cells in place.
Understanding this process is important because it’s the first step in metastasis, when cancer becomes more deadly and difficult to treat, said senior author David Sherwood, a biology professor at Duke.
For cancer cells to metastasize, they must first penetrate a dense sheet-like mesh of proteins and other molecules called the basement membrane, which surrounds tissues like a Kevlar wrapper.
But very few of the more than 200 anti-cancer drugs approved for clinical use actually prevent metastasis, the culprit behind 90% of cancer deaths.
Part of the problem is that the process has been hard to study. Cancer’s spread is unpredictable, and most cancer cells metastasize deep within the body, beyond the reach of light microscopes.
So Sherwood’s team studies a similar process in millimeter-long transparent worms called Caenorhabditis elegans.
During a worm’s development, a specialized cell called the anchor cell must break through the tough membrane that separates the worm’s uterus from its vulva to clear a path for the worm to lay eggs.
Both worm cells and human cancer cells rely on the same machinery to weaken and hack through tissue barriers. Enzymes help cut through the mesh, like molecular scissors. Tiny foot-like projections sprout from the cell surface and pound away, like cellular jackhammers. Invading cells also make stiff networks of actin filaments for a harder-hitting punch.
“There is a lot that needs to be made in the cell in order for invasion to occur,” said co-first author Isabel Kenny-Ganzert, a graduate student in Sherwood’s lab.
Sherwood and his team wanted to catalog the invasive cell’s toolkit.
In a new study published May 4 in the journal Development, co-author and Duke graduate student Daniel Costa tracked millions of gene readouts — what scientists call the transcriptome — produced within invading worm cells as they broke through the membrane.
The researchers identified 1,500 genes that are at least twice as active in invasive cells than in other cells in the worm’s body.
“This allows us to put together a profile of all the genes and proteins that seem to be important,” Kenny-Ganzert said.
“It’s basically a ‘parts list’ of what a cell needs to invade,” Sherwood said.
Using this list, the researchers identified many subtle drivers of cell invasion that had been missed by previous screens. The team was able to identify 13 genes that, when silenced, didn’t stop cells from breaking through, but did slow them down.
Another set of genes that were particularly active were genes that encode the building blocks of ribosomes, tiny machines that make the proteins that cells use to do their work.
The study showed that in the three or four hours leading up to invasion, the cell’s ribosome levels surge. Without them, the researchers found, invasive cells are cut off from the things they need. Proteins critical for invasion aren’t produced and cells can’t break through.
By attaching a glowing tag to the ribosomes, the researchers were also able to watch these cellular “protein factories” as they went about their business. In contrast to neighboring cells, whose ribosomes might be floating in the cell’s soupy interior, the ribosomes of invading cells were concentrated in an organelle called the endoplasmic reticulum, which is where proteins are synthesized and packaged for transport.
It’s a cell’s way of stepping up production prior to full-scale invasion, Sherwood said. “Kind of like upregulating the supply chain of an army so that it can send soldiers and equipment to the front line for battle.”
Although many cancers can be cured, the disease still causes one in six deaths each year worldwide, making it the second leading cause of death behind heart disease.
By studying the tools and tricks cells use to invade, the researchers hope to identify more effective ways to put the brakes on cancer’s spread.
“Most biological processes are like the Golden Gate Bridge, Sherwood said. “If you cut one of the steel cables, it’s still going to hold up. You have to cut a bunch of them before you see a bridge failure.”
Cell invasion is no different. “Most of the genes that are participating in invasion aren’t functioning alone,” Kenny-Ganzert said.
The researchers hope their work could make it possible to identify combinations of genes that, when knocked down together, might limit ribosome production and disrupt cell invasion’s supply chain.
This research was supported by grants from the National Institutes of Health (R35GM118049, R21OD028766, R21OD032430, R01GM121597, 5R35GM133573, R35GM134838, R35GM142880, T32CA009156 and R35GM142880).
CITATION: “The C. elegans Anchor Cell Transcriptome: Ribosome Biogenesis Drives Cell Invasion Through Basement Membrane,” Daniel S. Costa, Isabel W. Kenny-Ganzert, Qiuyi Chi, Kieop Park, Laura C. Kelley, Aastha Garde, David Q. Matus, Junhyun Park, Shaul Yogev, Bob Goldstein, Theresa V. Gibney, Ariel M. Pani, David R. Sherwood. Development, May 4, 2023. DOI: 10.1242/dev.201570
Journal
Development
DOI
10.1242/dev.201570
Method of Research
Experimental study
Subject of Research
Animals
Article Title
The C. elegans Anchor Cell Transcriptome: Ribosome Biogenesis Drives Cell Invasion Through Basement Membrane
Article Publication Date
4-May-2023



