Mechanistic approaches to chemically modify neurotoxicity of Alzheimer’s disease
Credit: KAIST
It has become evident recently that the interactions between copper and amyloid-β neurotoxically impact the brain of patients with Alzheimer’s disease. KAIST researchers have reported a new strategy to alter the neurotoxicity in Alzheimer’s disease by using a rationally designed chemical reagent.
This strategy, developed by Professor Mi Hee Lim from the Department of Chemistry, can modify the coordination sphere of copper bound to amyloid-β, effectively inhibiting copper’s binding to amyloid-β and altering its aggregation and toxicity. Their study was featured in PNAS last month.
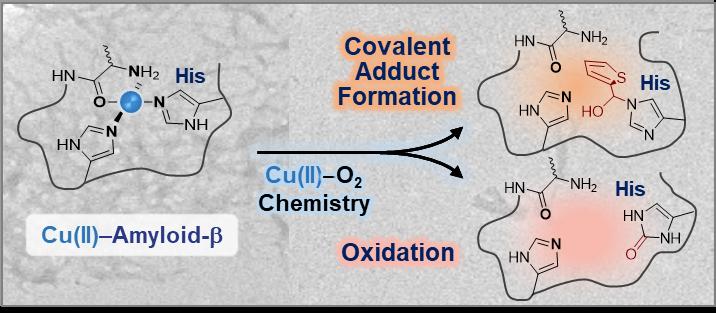
The researchers developed a small molecule that is able to directly interact with the coordination sphere of copper-amyloid-β? complexes followed by modifications via either covalent conjugation, oxidation, or both under aerobic conditions. The research team simply utilized copper-dioxygen chemistry to design a chemical reagent.
Answering how peptide modifications by a small molecule occur remains very challenging. The system includes transition metals and amyloidogenic proteins and is quite heterogeneous, since they are continuously being changed. It is critical to carefully check the multiple variables such as the presence of dioxygen and the type of transition metal ions and amyloidogenic proteins in order to identify the underlying mechanisms and target specificity of the chemical reagent.
The research team employed various biophysical and biochemical methods to determine the mechanisms for modifications on the coordination sphere of copper-A? complexes. Among them, peptide modifications were mainly analyzed using electrospray ionization-mass spectrometry.
Mass spectrometry (MS) has been applied to verify such peptide modifications by calculating the shift in exact mass. The research team also performed collision-induced dissociation (CID) of the target ion detected by MS to pinpoint which amino acid residue is specifically modified. The CID fragmentizes the amide bond located between the amino acid residues. This fragmental analysis allows us to identify the specific sites of peptide modifications.
The copper and amyloid-β complexes represent a pathological connection between metal ions and amyloid-β in Alzheimer’s disease. Recent findings indicate that copper and amyloid-β can directly contribute toward neurodegeneration by producing toxic amyloid-β oligomers and reactive oxygen species.
Professor Lim said, “This study illustrates the first experimental evidence that the 14th histidine residue in copper-amyloid-β? complexes can be specifically modified through either covalent conjugation, oxidation, or both. Considering the neurotoxic implications of the interactions between copper and amyloid-β, such modifications at the coordination sphere of copper in amyloid-β? could effectively alter its properties and toxicity.”
“This multidisciplinary study with an emphasis on approaches, reactivities, and mechanisms looks forward to opening a new way to develop candidates of anti-neurodegenerative diseases,” she added. The National Research Foundation of Korea funded this research.
###
-Profile
Mi-Hee Lim
Professor
[email protected]
http://sites.
Department of Chemistry
KAIST
-About KAIST
KAIST is the first and top science and technology university in Korea. KAIST was established in 1971 by the Korean government to educate scientists and engineers committed to industrialization and economic growth in Korea.
Since then, KAIST and its 64,739 graduates have been the gateway to advanced science and technology, innovation, and entrepreneurship. KAIST has emerged as one of the most innovative universities with more than 10,000 students enrolled in five colleges and seven schools including 1,039 international students from 90 countries.
On the precipice of semi-centennial anniversary in 2021, KAIST continues to strive to make the world better through the pursuit in education, research, entrepreneurship, and globalization.
Media Contact
Younghye Cho
[email protected]
82-423-502-294
Original Source
http://news.
Related Journal Article
http://dx.




