Credit: Reproduced with permission © 2016 American Chemical Society.
Tiny clusters of silver atoms arranged with atomic level precision could become more versatile and useful due to a simpler way to hold them together.
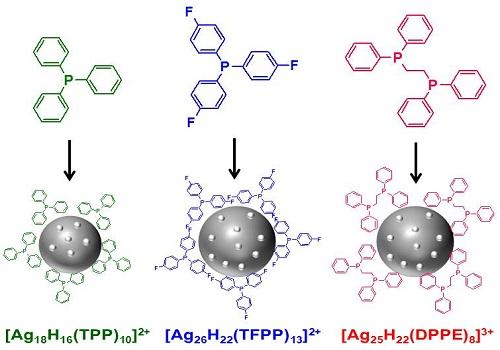
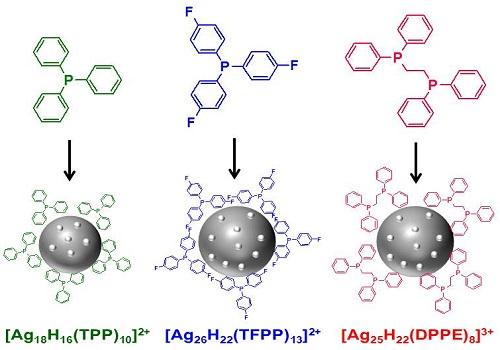
The nanoclusters assemble with the assistance of carbon-based organic ligands that form a shell around the metal atoms.
"Until now, most ligands have been big molecules that bond really strongly to the cluster and make its surface inaccessible to other chemical species," explained Osman Bakr, KAUST associate professor of material science and engineering. The smaller ligands developed at KAUST open new opportunities in nanocluster design and should broaden the chemical uses for such clusters1.
The ligands developed by Bakr and his team are known as hydrides because they have negatively charged hydrogen atoms (or hydride ions) in direct contact with the metal surface. The tiny hydride ions interact with small phosphorus-containing organic molecules known as phosphines to create a shell that protects and stabilizes the cluster.
Crucially, nanoclusters crystallize readily, allowing their structure to be precisely analyzed. Using this structural analysis to experiment with ligand design allows the properties of a nanocluster to be modified with atom-by-atom control.
One very promising potential application for nanoclusters is their use as catalysts to accelerate specific chemical reactions. The smaller ligands allow greater accessibility that widens the scope for reacting chemicals to reach the nanocluster's catalytic surface. The increased availability of that surface also allows other molecules to be deliberately added to assist with catalysis.
To design new catalysts, researchers need to know more about how they work, which is a major motivation for the KAUST team's work.
"These atomically precise nanoclusters are the key to understanding catalysis because, as we alter their size and shape, we can unlock the way in which catalysis occurs," said Bakr.
Because the electrons of the metal atoms are shared across the entire cluster, they also have unique optical and electronic properties. This allows the clusters to absorb light of many wavelengths and to remain in an energetically excited state for a long time.
Bar's group's members are pioneers in giving silver nanoclusters properties that were previously only seen in the much more expensive clusters made of gold. This could be vital for transferring research findings into economically viable applications.
Bakr noted, "The next challenge is to see if we can apply these innovations to other metals and greatly widen their usefulness."
###
Media Contact
Michelle D'Antoni
[email protected]
http://kaust.edu.sa/
############
Story Source: Materials provided by Scienmag