Background
1. Scientists from A*STAR's Singapore Immunology Network (SIgN) and KK Women's and Children's Hospital (KKH) in Singapore have discovered that a fetus's immune system is established as early as the second trimester of pregnancy, and may be able to initiate immune responses independently of the mother's immune system.
2. These findings debunk commonly-held assumptions about fetal immunity, including the idea that the fetus's immune system develops much later in the pregnancy cycle, and is unable to mount an independent immune response, being dependent on the mother's immune system to defend itself against external pathogens.
3. The scientists also found that an unborn baby's immune system contains a unique mechanism to prevent rejection of the mother's cells, even as it develops independently. This mechanism is mediated by dendritic cells expressing the protein Arginase-2.
4. These findings represent a landmark shift in our understanding of human immune development, and also provide insights into the immune mechanisms involved in some pregnancy-related illnesses and developmental diseases. 5. The findings of this study have been published in the scientific journal Nature on 14 June 2017.
Key Findings
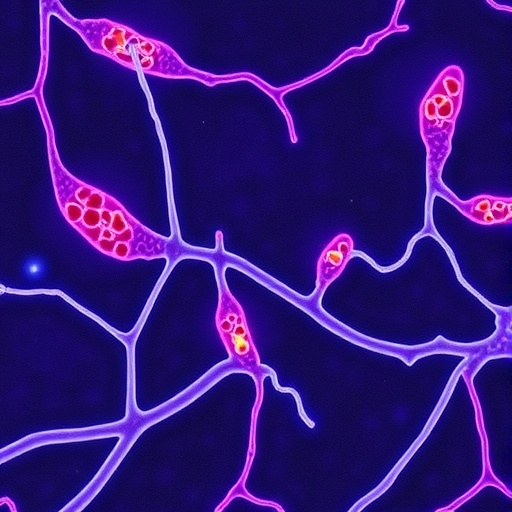
6. Dendritic cells are immune cells that act as the sentinels of the body's immune system. They detect and assess the threats posed by foreign pathogens to the body and decide whether to initiate an immune response.
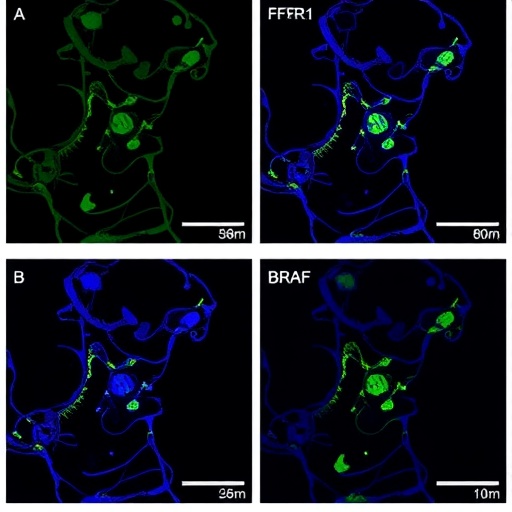
7. A major finding of the study is that as early as the second trimester of pregnancy, the human fetus has developed a network of dendritic cells that is similar to that of mature adults. These fetal dendritic cells are able to perform key functions associated with adult dendritic cells.
8. However, the scientists also uncovered a key difference between fetal and adult dendritic cells – the former expresses high levels of a protein called Arginase-2, which dampens the immune system response. This mechanism of immunosuppression promoted by fetal dendritic cells helps to ensure that the fetus does not reject the mother's cells even as it begins to develop its own immune defences.
9. Furthermore, Arginase-2 regulates the ability of immune cells to secrete a key inflammatory signaling protein called TNFα. This prevents the fetus's immune system from overreacting, and initiating unwanted inflammatory immune responses that could impact the baby's ongoing development in the womb.
10. The dysregulation of TNFα production in the fetal immune system has been implicated in various pregnancy-related conditions and developmental diseases, such as gestational diabetes mellitus, recurrent spontenous miscarriage, and necrotising enterocolitis. Hence, in expressing Arginase-2, fetal dendritic cells play a critical role in fetal immune tolerance, and ensuring the overall healthy development of the fetus in the mother's womb.
###
About the Agency for Science, Technology and Research (A*STAR)
The Agency for Science, Technology and Research (A*STAR) is Singapore's lead public sector agency that spearheads economic oriented research to advance scientific discovery and develop innovative technology. Through open innovation, we collaborate with our partners in both the public and private sectors to benefit society.
As a Science and Technology Organisation, A*STAR bridges the gap between academia and industry. Our research creates economic growth and jobs for Singapore, and enhances lives by contributing to societal benefits such as improving outcomes in healthcare, urban living, and sustainability. We play a key role in nurturing and developing a diversity of talent and leaders in our Agency and Research Institutes, the wider research community and industry. A*STAR oversees 18 biomedical sciences and physical sciences and engineering research entities primarily located in Biopolis and Fusionopolis. For more information on A*STAR, please visit http://www.a-star.edu.sg.
About A*STAR's Singapore Immunology Network (SIgN)
The Singapore Immunology Network (SIgN), officially inaugurated on 10 February 2006, is a research consortium under the Agency for Science, Technology and Research (A*STAR)'s Biomedical Research Council. The mandate of SIgN is to advance human immunology research and participate in international efforts to combat major health problems. Since its launch, SIgN has grown rapidly and currently includes 200 scientists from 25 different countries around the world working under 18 renowned Principal Investigators. At SIgN, researchers investigate immunity during infection and various inflammatory conditions including cancer and are supported by cutting edge technological research platforms and core services. Through this, SIgN aims to build a strong platform in basic human immunology research for better translation of research findings into clinical applications. SIgN also sets out to establish productive links with local and international institutions, and encourage the exchange of ideas and expertise between academic, industrial and clinical partners and thus contribute to a vibrant research environment in Singapore. For more information about SIgN, please visit http://www.a-star.edu.sg/sign.
About KK Women's and Children's Hospital (KKH)
KK Women's and Children's Hospital (KKH) is a recognised leader and Singapore's largest tertiary referral centre for Obstetrics, Gynaecology, Paediatrics and Neonatology. Founded in 1858, the 830-bed academic medical institution leads in patient-centred management of high risk conditions in women and children. More than 500 specialists adopt a compassionate, multi-disciplinary and holistic approach to treatment, and harness medical innovations and technology to deliver the best medical care possible. Accredited as an Academic Medical Centre, KKH is a major teaching hospital for all three medical schools in Singapore, Duke-NUS Medical School, Yong Loo Lin School of Medicine and Lee Kong Chian School of Medicine. The Hospital also runs the largest specialist training programme for Obstetrics and Gynaecology and Paediatrics in the country. Both programmes are accredited by the Accreditation Council for Graduate Medical Education International (ACGME-I), and are highly rated for the high quality of clinical teaching and the commitment to translational research. For more information, please visit http://www.kkh.com.sg.
ANNEX A: Quotes from Spokespersons:
Quote from Dr Florent Ginhoux, Senior Principal Investigator at SIgN, A*STAR, and joint-senior author of the study: "We are excited by the prospects this discovery holds for the growing field of research into fetal immunity, and the insights it provides into the very beginnings of the human immune system. Moving forward, we plan to expand our characterisation of the human fetal immune system beyond dendritic cells, to incorporate other key immune cells like B-cells or T-cells. Our eventual goal is to build an atlas of the fetal immune system. Hopefully, this will allow us to discover additional mechanisms of fetal tolerance, and identify gene signatures so that we can better evaluate fetal fitness and immunity."
Quote from Associate Professor Jerry Chan, Senior Consultant, Department of Reproductive Medicine, at KK Women's and Children's Hospital, Singapore, and joint-senior author of the study: "We have known for a long time that maternal cells cross into the baby's blood stream, but are not rejected by the fetal immune system. It had been long thought that the fetal immune system does not reject these semi-foreign cells due to its immaturity. The discovery of a fully functional dendritic cell network by the second trimester of pregnancy challenges this belief, and the high expression of Arginase-2 by fetal dendritic cells may contribute to the ability of the fetus to tolerate the mother's cells. This new insight lays the foundation for future immune-directed therapies, and contributes to our knowledge of the fetal origins of certain pregnancy-associated conditions, such as pre-eclampsia."
Notes for Editor:
The research findings described in this media fact sheet can be found in the 14th June 2017 online issue of the scientific journal Nature, under the title, "Human fetal dendritic cells promote pre-natal T cell immune-suppression through arginase-2" by Naomi McGovern1, Amanda Shin1,2, Gillian Low1, Donovan Low1, Kaibo Duan1, Leong Jing
Yao3, Rasha Msallam1, Ivy Low1, Nurhidaya Binte Shadan1, Hermi R Sumatoh1, Erin Soon1, Josephine Lum1, Esther Mok1, Sandra Hubert1, Peter See1, Edwin Huang Kunxiang4, Yie Hou Lee5,6, Baptiste Janela1, Mahesh Choolani7,8, Citra Nurfarah Zaini Mattar7,8, Yiping Fan4,8, Tony Kiat Hon Lim9, Dedrick Kok Hong9, Ker-Kan Tan10,11, John Kit Chung Tam11, Christopher Schuster12, Adelheid Elbe-Bürger12, Xiao-nong Wang13, Venetia Bigley13, Matthew Collin13, Muzlifah Haniffa13, Andreas Schlitzer1,14,15, Michael Poidinger1, Salvatore Albani3, Anis Larbi1, Evan W Newell1, Jerry Kok Yen Chan1,4,8,16 and Florent Ginhoux1.
1 Singapore Immunology Network (SIgN), A*STAR, 8A Biomedical Grove, Immunos Building, Level 3 and 4, Singapore
2 Shanghai Institute of Immunology, Shanghai Jiao Tong University School of Medicine, Shanghai, 200025, China
3 SingHealth Translational Immunology and Inflammation Centre (STIIC), 20 College Road, the Academia, Level 8 Discovery Tower, Singapore
4 Department of Reproductive Medicine, KK Women's and Children's Hospital, Singapore
5 KK Research Centre, KK Women's and Children's Hospital, 100 Bukit Timah Road, Singapore
6 OBGYN-Academic Clinical Program, Duke-NUS, Duke-NUS Medical School, 8 College Road, Singapore
7 epartment of Obstetrics & Gynaecology, Yong Loo Lin School of Medicine, National University of Singapore, NUHS Tower Block, 1E Kent Ridge Road, Singapore
8 Experimental Fetal Medicine Group, Yong Loo Lin School of Medicine, National University of Singapore, Singapore
9 Department of Pathology, Singapore General Hospital, 20 College Road, Singapore
10 Division of Colorectal Surgery, University Surgical Cluster, National University Health System,Singapore
11 Department of Surgery, Yong Loo Lin School of Medicine, National University of Singapore,1E Kent Ridge Road, Singapore
12 Department of Dermatology, DIAID, Medical University of Vienna, Währinger Gürtel 18-20, 1090 Vienna, Austria
13 Institute of Cellular Medicine, Newcastle University, Newcastle upon Tyne, United Kingdom
14 Myeloid Cell Biology, Life and Medical Science Institute, University of Bonn, 53115 Bonn Germany
15 Single Cell Genomics and Epigenomics Unit at the German Center for Neurodegenerative Diseases and the University of Bonn, 53175 Bonn,
Germany 16 Cancer and Stem Cell Biology Program, Duke-NUS Graduate Medical School, Singapore
Media Contact
Ms. Angeline Chen
[email protected]
65-639-42321
http://www.singhealth.com.sg/
############
Story Source: Materials provided by Scienmag