Brain tumours might arise when tissue does not heal properly– a finding that opens up new ideas about how cancer develops and how to combat it
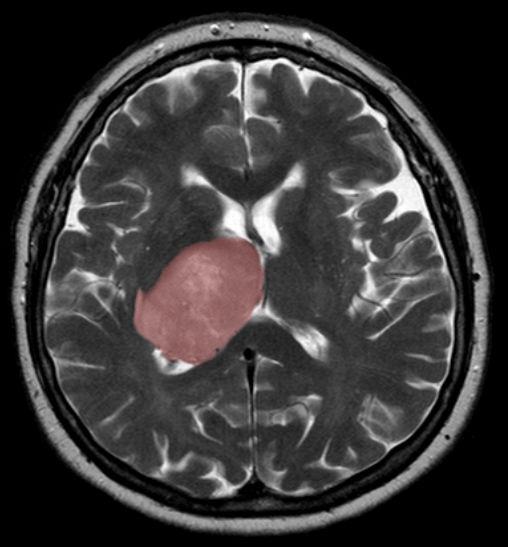
Credit: Hellerhoff, Wikimedia Commons
The healing process that follows a brain injury could spur tumour growth when new cells generated to replace those lost to the injury are derailed by mutations, Toronto scientists have found. A brain injury can be anything from trauma to infection or stroke.
The findings were made by an interdisciplinary team of researchers from the University of Toronto, The Hospital for Sick Children (SickKids) and the Princess Margaret Cancer Centre who are also on the pan-Canadian Stand Up To Cancer Canada Dream Team that focuses on a common brain cancer known as glioblastoma.
“Our data suggest that the right mutational change in particular cells in the brain could be modified by injury to give rise to a tumour,” says Dr. Peter Dirks, Dream Team leader who is the Head of the Division of Neurosurgery and a Senior Scientist in the Developmental and Stem Cell Biology program at SickKids.
Gary Bader, a professor of molecular genetics in the Donnelly Centre for Cellular and Biomolecular Research at U of T’s Temerty Faculty of Medicine and Dr. Trevor Pugh, Senior Scientist at the Princess Margaret, also led the research which has been published today in the journal Nature Cancer.
The findings could lead to new therapy for glioblastoma patients who currently have limited treatment options with an average lifespan of 15 months after diagnosis.
“Glioblastoma can be thought of as a wound that never stops healing,” says Dirks. “We’re excited about what this tells us about how cancer originates and grows and it opens up entirely new ideas about treatment by focusing on the injury and inflammation response.”
The researchers applied the latest single-cell RNA sequencing and machine learning technologies to map the molecular make-up of the glioblastoma stem cells (GSCs), which Dirks’ team previously showed are responsible for tumour initiation and recurrence after treatment.
They found new subpopulations of GSCs which bear the molecular hallmarks of inflammation and which are comingled with other cancer stem cells inside patients’ tumours. It suggests that some glioblastomas start to form when the normal tissue healing process, which generates new cells to replace those lost to injury, gets derailed by mutations, possibly even many years before patients become symptomatic, Dirks said.
Once a mutant cell becomes engaged in wound healing, it cannot stop multiplying because the normal controls are broken and this spurs tumour growth, according to the study.
“The goal is to identify a drug that will kill the glioblastoma stem cells,” says Bader, whose graduate student Owen Whitley contributed to the computational data analysis “But we first needed to understand the molecular nature of these cells in order to be able to target them more effectively.”
The team collected GSCs from 26 patients’ tumours and expanded them in the lab to obtain sufficient numbers of these rare cells for analysis. Almost 70,000 cells were analyzed by single-cell RNA sequencing which detects what genes are switched on in individual cells, an effort led by Laura Richards, a graduate student in Pugh’s lab.
The data confirmed extensive disease heterogeneity, meaning that each tumour contains multiple subpopulations of molecularly distinct cancer stem cells, making recurrence likely as existing therapy can’t wipe out all the different subclones.
A closer look revealed that each tumour has either of the two distinct molecular states–termed “Developmental” and “Injury Response”– or somewhere on a gradient between the two.
The developmental state is a hallmark of the glioblastoma stem cells and resembles that of the rapidly dividing stem cells in the growing brain before birth.
But the second state came as a surprise. The researchers termed it “Injury Response” because it showed an upregulation of immune pathways and inflammation markers, such as interferon and TNFalpha, which are indicative of wound healing processes.
These immune signatures were only picked up thanks to the new single-cell technology after being missed by older methods for bulk cell measurements.
Meanwhile, experiments led by Stephane Angers’ lab at the Leslie Dan Faculty of Pharmacy established that the two states are vulnerable to different types of gene knock outs, revealing a swathe of therapeutic targets linked to inflammation that had not been previously considered for glioblastoma.
Finally, the relative comingling of the two states was found to be patient-specific, meaning that each tumour was biased either toward the developmental or the injury response end of the gradient. The researchers are now looking to target these biases for tailored therapies.
“We’re now looking for drugs that are effective on different points of this gradient”, says Pugh, who is also the Director of Genomics at the Ontario Institute for Cancer Research. “There’s a real opportunity here for precision medicine– to dissect patients’ tumours at the single cell level and design a drug cocktail that can take out more than one cancer stem cell subclone at the same time.”
###
About The Hospital for Sick Children (SickKids)
The Hospital for Sick Children (SickKids) is recognized as one of the world’s foremost paediatric health-care institutions and is Canada’s leading centre dedicated to advancing children’s health through the integration of patient care, research and education. Founded in 1875 and affiliated with the University of Toronto, SickKids is one of Canada’s most research-intensive hospitals and has generated discoveries that have helped children globally. Its mission is to provide the best in complex and specialized family-centred care; pioneer scientific and clinical advancements; share expertise; foster an academic environment that nurtures health-care professionals; and champion an accessible, comprehensive and sustainable child health system. SickKids is a founding member of Kids Health Alliance, a network of partners working to create a high quality, consistent and coordinated approach to paediatric health care that is centred around children, youth and their families. SickKids is proud of its vision for Healthier Children. A Better World.
Media contact:
Jessamine Luck
Senior Communications Advisor
The Hospital for Sick Children (SickKids)
[email protected]
About Donnelly Centre for Cellular and Biomolecular Research
The Donnelly Centre for Cellular and Biomolecular Research at the University of Toronto is a globally renowned interdisciplinary research institute where scientists make discoveries to improve human health. Founded in 2005, the Centre currently houses 29 faculty members and over 300 students and postdoctoral trainees in the areas of integrative and systems biology, bioengineering, regenerative medicine and models of disease. Supported by competitive federal, provincial as well as international and nonprofit funding grants, the Donnelly Centre investigators build and apply the latest technologies with innovative and sophisticated platforms in a highly collaborative environment. In 2019, the Centre launched the Accelerator for Donnelly Collaboration, a biotechnology incubator for startups and companies looking to translate the discoveries made in the Centre into tangible medical advances. For more information: https:/
Media contact:
Jovana Drinjakovic, PhD
Science Communications Officer
Donnelly Centre for Cellular and Biomolecular Research
University of Toronto
[email protected]
C: 416.543.7820
@DonnellyCentre
About Princess Margaret Cancer Centre
Princess Margaret Cancer Centre has achieved an international reputation as a global leader in the fight against cancer and delivering personalized cancer medicine. The Princess Margaret, one of the top five international cancer research centres, is a member of the University Health Network, which also includes Toronto General Hospital, Toronto Western Hospital, Toronto Rehabilitation Institute and the Michener Institute for Education at UHN. All are research hospitals affiliated with the University of Toronto. For more information: http://www.
Media contact:
Katie Sullivan
Senior Public Affairs Associate
Princess Margaret Cancer Centre, University Health Network
Phone: 416-676-3221 (cell)
Email: [email protected]
Media Contact
Jovana Drinjakovic
[email protected]




