Credit: CeMM Research Center for Molecular Medicine of the Austrian Academy of Sciences
Diet-related diseases like non-alcoholic fatty liver disease (NAFLD) are known to have a major inflammatory component. However, the molecular pathways linking diet-induced changes with inflammation remained elusive. In a new study, scientists at the CeMM Research Center for Molecular Medicine of the Austrian Academy of Sciences and the Medical University of Vienna identified crucial inflammatory processes in NAFLD. Moreover, the study published in Hepatology shows that malondialdehyde (MDA), a biomarker for oxidative stress, plays a key role in the development of NAFLD and can be neutralized by specific natural antibodies – a novel approach towards a potential therapy for the prevalent disease.
The combination of a nutrient rich, western diet and a lack of exercise is a lifestyle which can lead to serious health issues: Worldwide, the incidences of obesity, hypertension or insulin resistance are alarmingly high. As a consequence, risk of developing inflammation-associated diseases like type 2 diabetes, NAFLD and cardiovascular disease increased accordingly. However, the exact pathways that link the eating habits with the ensuing inflammation were so far not well understood.
The team of Christoph Binder, Professor of Atherosclerosis Research at the Medical University of Vienna and Principal Investigator at CeMM, in collaboration with Ronit Shiri-Sverdlov at the Maastricht University, Christoph Reinhardt at the University Medical Center of the Johannes Gutenberg University Mainz and the German Center for Cardiovascular Research Mainz was not only able to shed light on the biological processes that lead to the development of chronic inflammation upon a nutrient rich diet in mice. Moreover, the scientists found MDA to be a key player in hepatic inflammation which can be neutralized with natural antibodies. Their results were published in Hepatology (DOI: 10.1002/hep.28970).
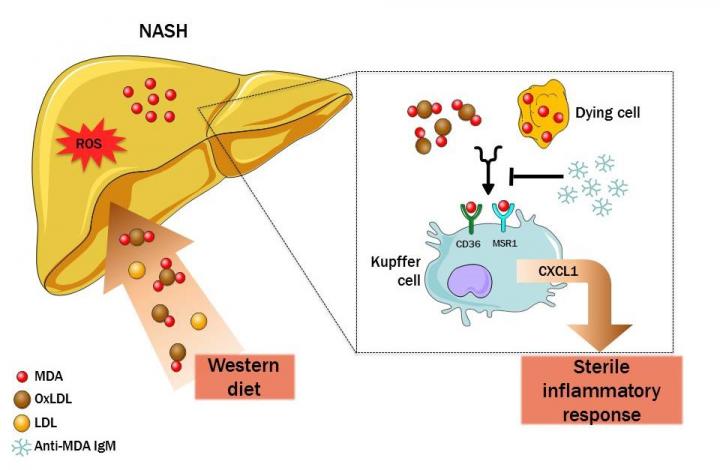
The highly reactive molecule malondialdehyde, a product from lipid decomposition and biomarker for oxidative stress, accumulates on the surface of dying cells in the liver. Chemically bound to membrane proteins or phospholipids, they form so called MDA epitopes – Binder's research group showed that those MDA epitopes induce cytokine secretion as well as leukocyte recruitment and thereby propagate existing inflammation and render it chronic.
"We were able to show in cell culture as well as in model organisms that those MDA epitopes play a major role in diet-induced hepatic inflammation," says Clara Jana-Lui Busch, one of the co-first authors of the study and PhD student at CeMM and the Medical University of Vienna. This was not the only insight of the study: "With intravenous injection of a specific MDA antibody which binds MDA epitopes selectively, we could ameliorate the inflammation in mice" Tim Hendrikx, the other co-first author and PostDoc in the group of Christoph Binder adds.
This study shows how the close collaboration of CeMM and the Medical University of Vienna fosters the development of a future precision medicine, says senior author Christoph Binder. "With cutting edge RNA sequencing methods and bioinformatic analyses of transcriptome data, we discovered key mechanisms in some of the most prevalent diseases and we confirmed those findings in mice models." Binder explains, and adds: "Above that, the administration of specific antibodies for MDA epitopes provide a promising new approach for the development of therapeutic strategies."
###
The study "Malondialdehyde epitopes are sterile mediators of hepatic inflammation in hypercholesterolemic mice" was published in Hepatology online in advance on December 16 2016. DOI: 10.1002/hep.28970
Authors: Clara Jana-Lui Busch*, Tim Hendrikx*, David Weismann, Sven Jäckel, Sofie M. A. Walenbergh, André F. Rendeiro, Juliane Weißer, Florian Puhm, Anastasiya Hladik, Laura Göderle, Nikolina Papac-Milicevic, Gerald Haas, Vincent Millischer, Saravanan Subramaniam, Sylvia Knapp, Keiryn L. Bennett, Christoph Bock, Christoph Reinhardt, Ronit Shiri-Sverdlov and Christoph J. Binder (*shared first author).
The study was funded by The Austrian Science Fund (FWF, SFB Lipotox F30), Boehringer Ingelheim (PhD Fellowship), Austrian Academy of Sciences (Doc Fellowship), EMBO (Short Term Fellowships), The Netherlands Organisation for Scientific Research (NWO), German Center for Cardiovascular Research (DZHK), German Federal Ministry of Education and Research, and the German Research Foundation (DFG).
Christoph Binder obtained his MD degree at the University of Vienna in 1997 and received his Ph.D. in Molecular Pathology at the University of California San Diego (UCSD) in 2002. Following a postdoctoral training period at UCSD, he joined the Institute for Laboratory Medicine of the Medical University of Vienna and became Principal Investigator at CeMM in 2006. In 2009 he was appointed Professor of Atherosclerosis Research at the Medical University of Vienna. Christoph Binder is dually affiliated with CeMM and the Medical University of Vienna. http://cemm.at/research/groups/christoph-binder-group/
The CeMM Research Center for Molecular Medicine of the Austrian Academy of Sciences is an interdisciplinary research institute committed to advancing the understanding of human diseases through basic and biomedical research. Located at the center of the Medical University of Vienna's campus, CeMM fosters a highly collaborative and interactive research mindset. Focusing on medically relevant questions, CeMM researchers concentrate on human biology and diseases like cancer and inflammation/immune disorders. In support of scientific pursuits and medical needs, CeMM provides access to cutting-edge technologies and has established a strategic interest in personalized medicine. Since 2005, Giulio Superti-Furga is the Scientific Director of CeMM. http://www.cemm.at
The Medical University of Vienna is one of the most traditional medical education establishments with nearly 8,000 students and approximately 5,500 staff members, and one of the most important top-level biomedical research institutions in Europe. Its international outlook is one of its most important pillars and the research focus is centered on immunology, cancer research, imaging, brain research and cardiovascular diseases. https://www.meduniwien.ac.at/web/en
For further information please contact
Mag. Wolfgang Däuble http://Media Relations Manager
CeMM
Research Center for Molecular Medicine
of the Austrian Academy of Sciences
Lazarettgasse 14, AKH BT 25.3
1090 Vienna, Austria
Phone +43-1/40160-70 057
Fax +43-1/40160-970 000
[email protected]
http://www.cemm.at
Media Contact
Wolfgang Däuble
[email protected]
43-140-160-70057
@CeMM_News
http://www.cemm.oeaw.ac.at
############
Story Source: Materials provided by Scienmag