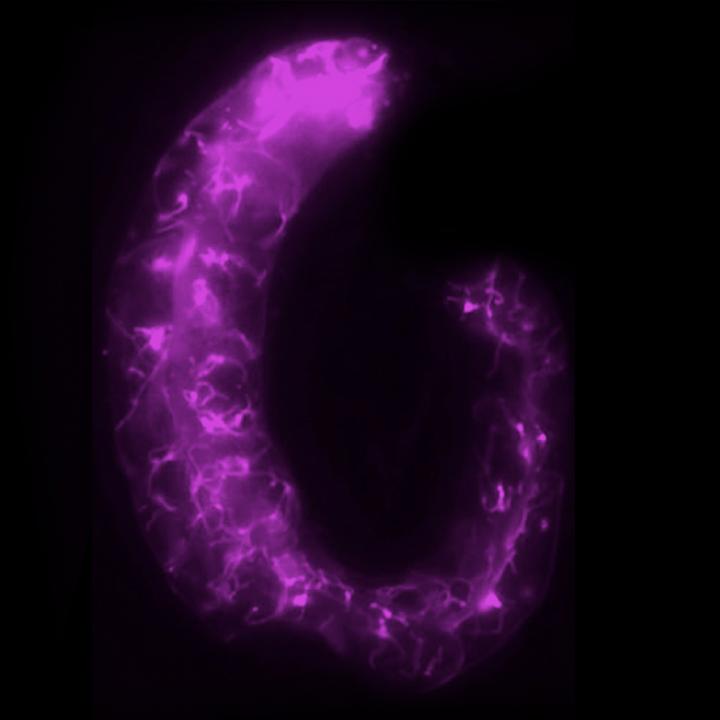
Credit: Alyssa Johnson and Adam Bohnert, LSU
Alyssa Johnson and Adam Bohnert, assistant professors in the Louisiana State University Department of Biological Sciences and experts in cell and molecular biology, have discovered a new class of lysosomes that they refer to as “tubular lysosomes.” This breakthrough could lead to medical therapies and treatments to slow–or even reverse–aging and disease in humans and animals. The researchers recently received a $1 million grant from the W. M. Keck Foundation to advance their work.
Lysosomes are generally known as cellular “garbage dumps” or recycling stations for obsolete and undesirable materials, including bacteria and viruses. Originally discovered in 1949, lysosomes have always been drawn as homogenous, spherical vesicles, not unlike SpaghettiOs. Johnson and Bohnert, meanwhile, have found that lysosomes can form complex, lattice-like networks, which can greatly affect the outcome of aging, disease and other stressors.
While Johnson mostly studies fruit flies (Drosophila) and Bohnert’s focus is on worms (C. elegans), they have also looked at mammalian cells. Their findings translate well between organisms. Not every tissue in every organism contains tubular lysosomes, however. Some never have them, while tubular lysosomes can be stimulated and made to grow in others. A starved worm, for example, will grow tubular lysosomes in certain tissues. Not only does this help the worm survive the temporary starvation; it may also help the worm be healthier and live longer after it returns to a normal diet. And, surprisingly, the worm’s progeny appear to benefit, too. The researchers are testing whether tubular lysosomes can be passed from a parent to a child and then onto grandchildren and so on.
In the short term, the researchers’ discovery could help explain the benefits of intermittent fasting. In the long term, their work could find ways to induce tubular lysosome activity in human and animal tissues to extend health and longevity for not just one individual, but for multiple generations.
“This is a major black box in biology where we don’t have a lot of knowledge yet,” Bohnert said. “From a very basic level, we’re now redefining the landscape of what it means to be a cell. Tubular lysosomes could hint at ways to slow down the aging process, or even reverse it. This sounds like science fiction, but it could be possible.”
Johnson was the first to observe tubular lysosomes in muscle tissue in a fruit fly.
“My first thought was, ‘This is beautiful!'” she recalls. “I had never before seen lysosomes that look like this in any cell type. At that moment, I knew in my heart that I had to study them further.”
“Alyssa and I complement each other because we have expertise in different model organisms; she in flies and me in worms,” Bohnert continued. “We’ve now developed a toolkit to go in and assess on a broad level any tubular lysosome. How does it act? How does it function? How is it built? How is it stimulated?”
So far, the researchers have identified a couple of genes that appear to control tubular lysosomes.
“One of the genes, if you over-express it, can make networks in places where there normally are no networks, and this seems to correlate with animal health,” Johnson explains. “Our question is, can we introduce it in animals and tissues that don’t normally have them, to promote their health? We’re trying to understand the differences between tissues and if we can learn from one tissue to provide beneficial output for a different tissue, or the whole organism in general.”
Worms have turned out to be a near-ideal animal to study tubular lysosomes in. Since C. elegans are transparent, it is easy for the researchers to watch the lattice of tubular lysosomes either grow and connect or shrink and disconnect as they look at whole, living animals under fluorescence microscopes.
The fact that the worms can be kept alive during imaging is an integral part of the discovery process–perhaps the main reason tubular lysosomes were unknown until now is that they only can be observed in live tissue.
“We’re really the only ones studying tubular lysosomes in the world, which puts us in a unique position to break open this field,” Johnson said. “There’s the basic, fundamental science part of our work and then the biomedically relevant part, where we use our new knowledge of lysosome biology to see how it could be used for therapeutic purposes. That’s what made it possible for us to get support from the Keck Foundation. We’ll do genome-wide screens, biochemical screens and broad analysis of tissues. We’ll find new genetic players that regulate and control these tubular lysosomes.”
Johnson and Bohnert don’t just work together; they are married with two kids. Recently, their son was gifted a coloring book where he could color cells. Inside, the lysosome looked like a SpaghettiO, just like in Johnson and Bohnert’s biology textbooks from college. Their ongoing research will now likely lead to revisions in both coloring books and textbooks, as it redefines our perception of life at its most basic unit–the cell.
“The differences we’ve seen in different tissues will be really important for future studies,” Johnson concludes. “Often, people study organelles or some function of organelles in one system or cell type and make assumptions about that organelle universally. But based on what we’ve seen, that’s really not true for the lysosome. In every tissue, lysosomes look and behave quite differently.”
###
The W. M. Keck Foundation was established in 1954 in Los Angeles by William Myron Keck, founder of The Superior Oil Company. One of the nation’s largest philanthropic organizations, the W. M. Keck Foundation supports outstanding science, engineering and medical research, and undergraduate education. The Foundation also maintains a program within Southern California to support arts and culture, education, health and community service projects and programs.
Media Contact
Alison Satake
[email protected]
225-578-3870
Original Source
https:/




