In a groundbreaking study that promises to reshape our understanding of Alzheimer’s disease (AD) at the molecular and cellular levels, researchers have unveiled a pivotal link between calcium dynamics in astrocytes and the malfunctioning of the brain’s glymphatic system. This discovery, emerging from a detailed investigation using the 5xFAD mouse model of AD—a well-established proxy for familial AD pathology—unveils how amyloid-β protein accumulation triggers a cascade of disruptions that compromise brain waste clearance, providing fresh insights into early disease mechanisms that could pave the way for novel therapeutic strategies.
The glymphatic system, composed primarily of astrocytic aquaporin-4 (AQP4) water channels, is crucial for the effective clearance of metabolic waste from the brain. Its function is tightly regulated by the polarity and distribution of AQP4 on astrocytic endfeet, facilitating cerebrospinal fluid exchange and removal of neurotoxic substances. However, in AD, the compromise of this system has been increasingly recognized as a major contributor to pathological progression, yet the precise molecular underpinnings remained enigmatic—until now.
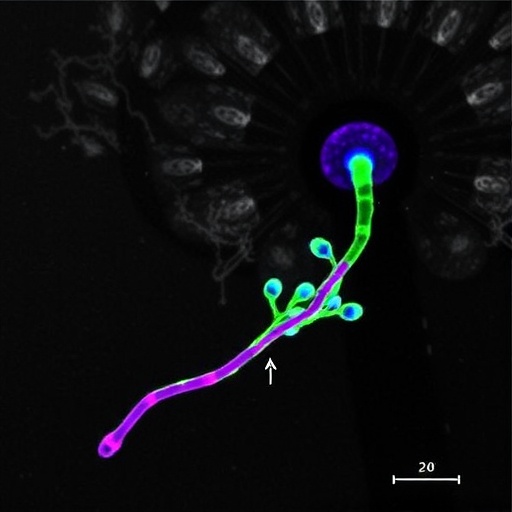
Central to this new research is the observation that astrocytes in the medial prefrontal cortex of 5xFAD mice exhibit heightened intracellular calcium (Ca²⁺) dynamics in response to amyloid-β exposure. This aberrant calcium signaling, driven particularly by Gq protein-coupled receptor (GPCR) activation, emerges as a critical upstream event, linking amyloid pathology to downstream glymphatic dysfunction. The findings reveal that elevated Ca²⁺ levels in astrocytes induce a marked increase in cholesterol biosynthesis, perturbing membrane composition and affecting protein trafficking within astrocytic processes.
Mechanistically, this cholesterol surge in astrocytes leads to enhanced endocytosis and lysosomal targeting of AQP4, effectively reducing its localization on the astrocytic endfeet where it is needed for water transport. This mislocalization disrupts AQP4 polarity, a phenomenon directly correlated with impaired glymphatic perfusion and waste clearance. The study meticulously charts how cholesterol-mediated AQP4 trafficking defects attenuate glymphatic efficiency, promoting the accumulation of neurotoxic waste products and facilitating cognitive decline observed in AD model mice.
Strikingly, the researchers demonstrated that targeted attenuation of Gq GPCR-evoked calcium signaling ameliorates these deficits. By pharmacologically or genetically dampening astrocytic calcium activity, the aberrant cholesterol synthesis and AQP4 mislocalization were significantly reduced, restoring glymphatic function. This link between calcium homeostasis and cholesterol metabolism in astrocytes offers an unprecedented mechanistic framework connecting amyloid-β pathology with glymphatic and cognitive dysfunction.
Moreover, the study explored therapeutic strategies focused on cholesterol biosynthesis modulation. Using either astrocyte-specific knockdown of squalene epoxidase, a key enzyme in cholesterol synthesis, or systemic administration of atorvastatin—a commonly prescribed cholesterol-lowering drug—the researchers successfully improved glymphatic perfusion and meningeal lymphatic drainage. These interventions not only restored AQP4 polarity but also yielded measurable cognitive benefits in 5xFAD mice, underscoring the translational potential of targeting astrocytic cholesterol metabolism in early AD.
This research adds a new dimension to the multifaceted landscape of AD pathogenesis by interlinking amyloid-β-induced astrocytic calcium dysregulation with membrane lipid remodeling and impaired protein trafficking. The demonstration that cholesterol synthesis can be modulated to restore the integrity of glymphatic and lymphatic coupling furthers the growing evidence that astrocytes play an active and dynamic role in neurodegenerative disease progression rather than being mere bystanders.
The implications of these findings extend beyond molecular neuroscience, suggesting that existing pharmacological agents targeting cholesterol pathways could be repurposed or optimized to mitigate AD-related glymphatic failure. Given the pressing need for early interventions in AD, therapies that preserve or enhance the brain’s natural waste clearance mechanisms represent a promising avenue for slowing disease progression and preserving cognitive function.
Importantly, this study also highlights the intricate balance of astrocytic calcium signaling in maintaining homeostatic brain functions. While calcium is a ubiquitous signaling molecule essential for multiple cellular processes, its dysregulation in AD appears to fuel pathological lipid metabolic changes that undermine cellular and network-level integrity. Future research might delve deeper into how calcium signaling intersects with other astrocytic pathways to orchestrate both healthy function and disease states.
The research team’s use of the 5xFAD mouse model, which recapitulates key features of human AD pathology including amyloid plaque accumulation and cognitive deficits, strengthens the translational relevance of their findings. Coupled with sophisticated imaging and molecular biology techniques, their multi-level approach elucidates a previously underappreciated astrocyte-centric mechanism that bridges amyloid pathology and brain clearance deficits.
Another important facet of this discovery is the reinforced conceptual model that views the glymphatic system as a critical therapeutic target in neurodegeneration. Prior studies have implicated impaired glymphatic clearance in a range of neurological disorders, but few have elucidated concrete molecular pathways. Here, the direct link from amyloid-β-induced Ca²⁺ dynamics to cholesterol-driven AQP4 trafficking provides a compelling biochemical axis for future drug development.
In addition to astrocytic calcium signaling and cholesterol metabolism, the findings spotlight the role of meningeal lymphatic vessels in maintaining brain health. Enhancing meningeal lymphatic drainage through cholesterol synthesis modulation not only improved waste clearance but also positively impacted cognitive outcomes in AD models. This underscores the importance of vascular and lymphatic crosstalk in CNS disorders.
These discoveries herald a potential shift toward combination therapies that target multiple hallmarks of Alzheimer’s pathology. By combining amyloid-lowering approaches with interventions that normalize astrocytic calcium and cholesterol homeostasis, there may be synergistic improvements in glymphatic function and neuroprotection, offering hope for more effective disease-modifying treatments.
As Alzheimer’s disease continues to pose an escalating global health challenge, the delineation of novel astrocyte-specific targets introduces fresh avenues for early diagnosis and intervention. Given the accessibility of cholesterol synthesis pathways to pharmacological modulation, the translation of these findings to clinical trials may hasten the development of therapies designed to uphold cerebral waste clearance and delay neurodegeneration.
Ultimately, the integration of astrocytic physiological mechanisms with the broader context of neural network function and systemic clearance pathways provides a richer understanding of AD pathophysiology. This will be crucial as the neuroscience community moves toward comprehensive models of disease that encompass cellular signaling, lipid metabolism, glial function, and vascular biology.
In conclusion, the discovery that amyloid-β-driven astrocytic calcium hyperactivity elevates cholesterol synthesis disrupting AQP4 localization and glymphatic flow offers exciting new insights into Alzheimer’s disease mechanisms. By intervening at the intersection of calcium signaling and membrane lipid homeostasis, researchers have illuminated promising therapeutic pathways capable of preserving brain clearance systems and cognitive integrity amidst neurodegeneration. This work not only broadens the horizon of AD research but also substantiates the critical role of astrocytes as central players in maintaining neural health.
Subject of Research: Astrocytic calcium dynamics, cholesterol metabolism, and glymphatic system dysfunction in Alzheimer’s disease.
Article Title: Amyloid-β-driven glymphatic dysfunction in Alzheimer’s disease model mice is driven by Ca²⁺-mediated increases in astrocytic cholesterol.
Article References: Zhang, Z., Li, S., Xu, L. et al. Amyloid-β-driven glymphatic dysfunction in Alzheimer’s disease model mice is driven by Ca²⁺-mediated increases in astrocytic cholesterol. Nat Neurosci (2026). https://doi.org/10.1038/s41593-026-02261-9
Image Credits: AI Generated
DOI: https://doi.org/10.1038/s41593-026-02261-9
Tags: 5xFAD mouse model familial Alzheimer’sAlzheimer’s disease glymphatic system dysfunctionamyloid-beta impact on astrocytesaquaporin-4 polarity disruption Alzheimer’sastrocyte cholesterol in brain waste clearanceastrocyte-mediated cerebrospinal fluid exchangeastrocytic calcium dynamics Alzheimer’s pathologybrain metabolic waste clearance mechanismscalcium signaling in astrocytes ADmolecular mechanisms of Alzheimer’s diseaseneurodegeneration and glymphatic impairmenttherapeutic targets