In a groundbreaking study set to redefine our understanding of psoriasis, researchers have uncovered a pivotal role for epidermal MHC class II molecules in orchestrating the recruitment of natural killer (NK) cells to the skin. This immunological interplay precipitates pyroptosis—a highly inflammatory form of programmed cell death—in keratinocytes, thereby fueling the pathological progression of psoriasis. This discovery sets a new paradigm in dermatological immunology, unraveling the complex cellular crosstalk that underpins this chronic and often debilitating skin condition.
Psoriasis, traditionally viewed through the lens of aberrant immune activation and keratinocyte proliferation, has long challenged scientists due to its multifaceted etiology. Historically, much emphasis was placed on T cell-mediated responses as the primary drivers of disease pathology. However, this recent investigation pivots the spotlight towards innate immune effectors—specifically NK cells—and their interaction with epidermal cells, mediated through the expression of MHC-II molecules. This novel axis reveals previously uncharted territories in psoriasis pathogenesis.
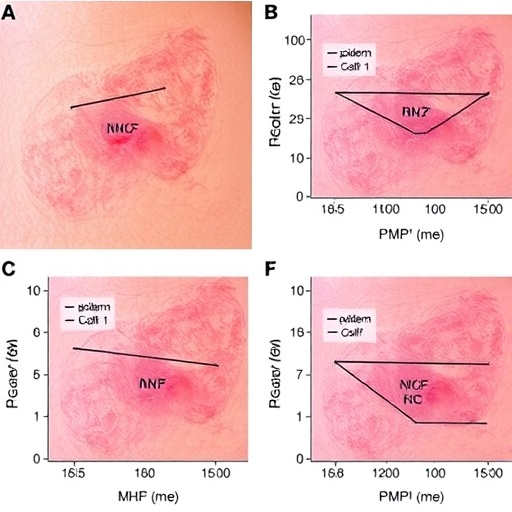
At the molecular heart of this mechanism lies the expression of MHC class II molecules on epidermal keratinocytes, a feature not typically associated with these cells under homeostatic conditions. The study demonstrates that during psoriatic inflammation, keratinocytes aberrantly upregulate MHC-II, effectively transforming them into atypical antigen-presenting cells. This upregulation serves as a beacon that recruits circulating NK cells to the epidermis, a process that has been meticulously delineated using sophisticated in vivo and in vitro models reflective of human disease.
The recruited NK cells, upon engagement with MHC-II-expressing keratinocytes, undergo activation that culminates in the secretion of cytotoxic mediators triggering pyroptotic cell death within the epidermis. Unlike apoptosis, pyroptosis is characterized by the formation of membrane pores, cellular swelling, and the release of pro-inflammatory cytokines such as IL-1β and IL-18. This cascade amplifies local inflammation, recruiting additional immune cells and exacerbating the psoriatic lesions, creating a feedback loop that sustains and escalates the disease state.
Mechanistically, the interaction between NK cells and keratinocytes hinges on receptor-ligand crosstalk facilitated by MHC-II molecules, which, in this context, appear to act beyond classic antigen presentation roles. The study uncovers that NK cell activation is contingent upon direct recognition of these MHC-II molecules, highlighting a noncanonical pathway wherein NK cells modulate keratinocyte fate through targeted pyroptosis. This insight challenges long-established immunological dogma and opens avenues for exploring NK cell behavior in other inflammatory skin diseases.
Translated from bench to bedside, these findings possess profound therapeutic implications. Targeting the MHC-II mediated recruitment of NK cells or intercepting the pyroptotic signaling cascade could ameliorate psoriatic inflammation, offering a precision medicine approach. Current psoriasis therapies largely focus on systemic immunosuppression or biologics directed at cytokines such as TNF-α and IL-17; however, this study advocates for a paradigm shift by tuning innate immunity at the epidermal interface.
Further, the identification of pyroptosis as a critical effector mechanism in keratinocyte demise underscores the broader significance of inflammatory cell death pathways in skin disease. It beckons the exploration of selective inhibitors of pyroptotic components such as gasdermin D, caspase-1, and inflammasome complexes, which might serve as novel pharmacological targets. Such interventions could diminish inflammatory amplification without broadly suppressing immune competence.
This research also exemplifies the complex cellular heterogeneity within psoriatic lesions, spotlighting keratinocytes not merely as passive victims of immune attack but as active participants in immune modulation. Their transformation into MHC-II-expressing entities suggests plasticity and adaptability in response to inflammatory cues, revolutionizing the way epidermal responses are conceptualized in chronic dermatoses.
Technical rigor was a hallmark of this study, employing cutting-edge techniques including single-cell RNA sequencing to delineate expression profiles of MHC-II and pyroptosis-related genes in keratinocytes. Coupled with high-resolution imaging and functional assays, the comprehensive approach furnished incontrovertible evidence linking epidermal MHC-II expression to NK cell-mediated pyroptosis, thus cementing this axis as a cornerstone in psoriasis pathobiology.
Moreover, animal models genetically engineered to abrogate MHC-II expression on keratinocytes exhibited markedly reduced NK cell infiltration and attenuated psoriatic pathology, reinforcing causality. This genetic evidence substantiates the mechanistic hypothesis, elevating the translational potential of targeting this pathway in human patients.
Significantly, the temporal dynamics of NK cell recruitment and subsequent pyroptosis induction appear tightly regulated and disease-stage dependent, suggesting potential windows for therapeutic intervention that could preempt irreversible skin damage. Understanding these kinetics further facilitates the design of treatment modalities that are both effective and minimally disruptive to normal skin homeostasis.
The study also invites reconsideration of NK cells’ roles beyond cytotoxicity, suggesting their function as regulatory sentinels shaping epidermal integrity through controlled cell death pathways. This dualistic role exemplifies the delicate balance of immune surveillance and tissue homeostasis, a balance disrupted in psoriatic disease states.
In addition, the data propose that aberrant epidermal MHC-II expression may serve as a biomarker for disease activity or severity in psoriasis, enabling clinicians to stratify patients and tailor interventions more precisely. This biomarker potential, combined with emerging therapeutic targets, gestures towards a future where personalized medicine optimally manages this chronic condition.
While illuminating, the findings also raise pivotal questions regarding the upstream signals inducing MHC-II expression on keratinocytes and the broader immunological milieu influencing NK cell behavior. Decoding these triggers will be essential to fully manipulate this pathway therapeutically and to understand its relevance across diverse inflammatory skin disorders.
Ultimately, this study by Yi, Yu, Wang and colleagues constitutes a watershed moment in psoriasis research, integrating molecular immunology, cell biology, and translational medicine. By delineating the epidermal MHC-II–NK cell–pyroptosis axis, it lays a robust foundation for innovative treatment strategies aimed at restoring skin integrity by modulating innate immune interactions.
As the prevalence of psoriasis continues to rise globally, unraveling such intricate cellular dialogues provides an invaluable beacon of hope. Future research inspired by these findings promises not only to refine therapeutic approaches but also to deepen our overarching grasp of skin immunology’s complexities, paving the way for breakthroughs that extend beyond psoriasis to other inflammatory and autoimmune conditions.
Subject of Research:
The study investigates the role of epidermal MHC class II molecules in mediating NK cell recruitment and triggering pyroptosis in keratinocytes, contributing to the pathogenesis of psoriasis.
Article Title:
Epidermal MHC-II-mediated NK cell recruitment triggers keratinocyte pyroptosis, facilitating pathogenesis of psoriasis.
Article References:
Yi, X., Yu, P., Wang, J. et al. Epidermal MHC-II-mediated NK cell recruitment triggers keratinocyte pyroptosis, facilitating pathogenesis of psoriasis. Exp Mol Med (2026). https://doi.org/10.1038/s12276-026-01717-z
Image Credits:
AI Generated
DOI:
https://doi.org/10.1038/s12276-026-01717-z
Tags: chronic skin inflammationepidermal MHC class II in psoriasisimmunological mechanisms of psoriasisinnate immunity in psoriasis pathogenesiskeratinocyte antigen presentationkeratinocyte programmed cell deathMHC-II upregulation in keratinocytesnatural killer cell recruitment in skinNK cell and keratinocyte interactionNK cell-mediated pyroptosisnovel targets for psoriasis therapypsoriasis immunopathology