Immune checkpoint inhibitors (ICIs) have revolutionized cancer therapeutics, offering remarkable benefits across multiple malignancies, including hepatocellular carcinoma (HCC). However, their expanding use has shone a spotlight on an increasingly recognized complication: immune-related hepatotoxicity, or checkpoint inhibitor‑induced liver injury (ChILI). As ICIs unleash the immune system against cancer, unintended immune attack on hepatic tissue can lead to serious liver damage. This emerging challenge has ignited intense debate and uncertainty over optimal diagnosis, management, and safe rechallenge protocols.
One of the most striking features of ChILI is its complex and variable presentation. The liver injury can manifest as predominantly hepatocellular damage, cholestatic injury, or mixed phenotypes. Underlying liver disease like cirrhosis complicates the clinical picture further, making it difficult to distinguish immunotherapy-induced damage from tumor progression or viral flares. Consequently, clinicians grapple with imprecise diagnostic criteria and divergent guideline recommendations from major societies such as ASCO, ESMO, SITC, AGA, and NCCN.
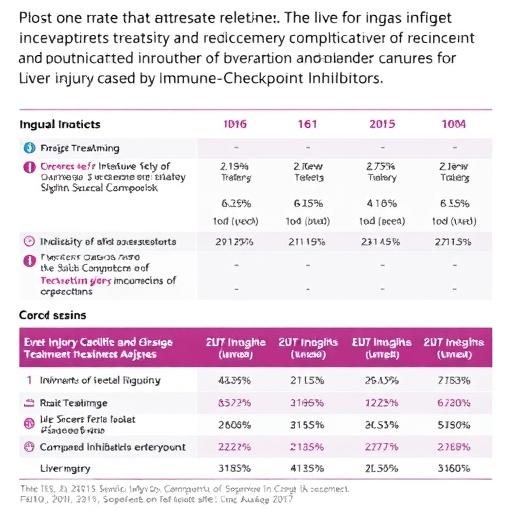
Current expert consensus broadly endorses corticosteroids as the frontline treatment for significant immune-related hepatotoxicity. Yet, controversy swirls around when to initiate steroids and at what threshold of liver enzyme elevation. Some bodies advocate early steroid commencement at grade 2 hepatotoxicity if no improvement occurs, while others recommend a more conservative approach. This lack of harmonization reflects the sparse high-quality evidence base and reliance on extrapolations from general immune-related adverse event management principles.
Despite steroids’ pivotal role, up to 30% of patients exhibit steroid-refractory liver injury. Mechanistically, this resistance may arise from severe T-cell mediated hepatocyte destruction, small-duct cholangitis patterns inducing ductopenia, delayed recognition with advanced inflammation, and interplay of alternative immune pathways. In these cases, escalation of immunosuppression becomes necessary, yet therapeutic options remain limited and under-studied. Mycophenolate mofetil (MMF) is the most common second-line agent employed, though its efficacy lacks robust randomized trial support.
Intriguingly, ursodeoxycholic acid (UDCA) has emerged as a potentially valuable adjunctive treatment, particularly in cholestatic patterns of ChILI. Retrospective analyses have shown that UDCA monotherapy yielded biochemical improvement in the majority of cholestatic patients, although some with macroscopic bile duct injury experienced notable recurrence rates. The combination of UDCA with corticosteroids appears to enhance safety and efficacy, underscoring the need for personalized therapeutic strategies tailored to histopathological injury types.
When it comes to tissue diagnosis, the role of liver biopsy remains controversial but crucial, especially in patients with atypical, prolonged hepatitis or underlying cirrhosis. Some guidelines consider biopsy optional, whereas others strongly recommend it to elucidate injury patterns and exclude differential diagnoses such as tumor infiltration or viral hepatitis reactivation. This is particularly vital in HCC patients, where overlapping pathologies coexist, challenging the clinician’s ability to time immunosuppressant initiation appropriately.
The question of rechallenging patients with ICIs after resolution of ChILI is rapidly gaining clinical relevance, given the substantial survival benefits ICIs confer in advanced malignancies. Retrospective and prospective studies collectively report recurrence rates of immune-related hepatotoxicity upon rechallenge ranging from 20 to 35%. Most relapses are fortunately mild and manageable, with no fatal outcomes reported to date. Importantly, recurrence tends to be more frequent in patients with prior cholestatic injury or those harboring autoimmune markers such as elevated antinuclear antibodies.
Emerging data suggest that patients who experience recurrence upon rechallenge may paradoxically enjoy better oncologic outcomes, hinting at an intricate interplay between immune activation, toxicity, and tumor control. Nevertheless, guidelines diverge significantly: some endorse cautious rechallenge post-resolution of grade 2 toxicity, others are more conservative, particularly after severe (grade ≥3) liver injury or second-line immunosuppression. NCCN favors discontinuation only at life-threatening grade 4 toxicity, highlighting the need for individualized, risk-benefit analyses.
Differences in international recommendations underscore the profound gaps in evidence guiding management of immune-related hepatotoxicity. Real-world data suggest higher rates of steroid-refractory ChILI and complexities in distinguishing true toxicity from tumor biology, particularly in cirrhotic HCC patients. Multidisciplinary evaluation coupled with histological confirmation enhances diagnostic accuracy and informs treatment, but standardized algorithms remain elusive.
Looking forward, the liver-oncological community must prioritize well-designed prospective studies evaluating optimal immunosuppressive regimens, ideal steroid tapering schedules, and the efficacy of novel agents like MMF and UDCA. Crucially, harmonized criteria for biopsy and rechallenge are imperative to optimize patient outcomes. Such evidence-based frameworks would mitigate current controversies and provide a rational, patient-specific approach to balancing toxicity management with cancer control.
In sum, immune checkpoint inhibitor-induced liver injury represents a multifaceted challenge at the intersection of immunology, hepatology, and oncology. As these agents continue to transform cancer care paradigms, refining diagnostic tools, elucidating pathogenetic mechanisms, and harmonizing management protocols will be vital. With careful risk stratification and multidisciplinary collaboration, rechallenge therapy may unlock extended survival benefits without compromising safety, heralding a new era in treating immune-related hepatotoxicity.
Subject of Research: Immune checkpoint inhibitor-induced liver injury (ChILI) and its management controversies
Article Title: Immune Checkpoint Inhibitor–induced Liver Injury: A Critical Appraisal of Treatment and Rechallenge Controversies
News Publication Date: 19-Jan-2026
Web References: http://dx.doi.org/10.14218/JCTH.2025.00450
Keywords: immune checkpoint inhibitors, hepatotoxicity, liver injury, corticosteroids, ursodeoxycholic acid, immune-related adverse events, hepatocellular carcinoma, immunotherapy rechallenge, mycophenolate mofetil, liver biopsy, immune-mediated hepatitis
Tags: checkpoint inhibitor-induced hepatotoxicity diagnosiscorticosteroid timing in liver injurycorticosteroid treatment for ChILIdistinguishing ChILI from liver disease progressionguidelines for immune-related liver toxicityhepatocellular carcinoma immunotherapy complicationsimmune checkpoint inhibitor liver injuryimmune checkpoint inhibitors adverse effectsimmune-related hepatotoxicity managementliver injury rechallenge protocolsmanaging immune checkpoint inhibitor hepatotoxicityvariable presentations of immune checkpoint inhibitor liver damage