In the relentless quest for innovative cancer therapies, natural compounds have continuously emerged as a promising frontier. Over the past two decades, biologically active polysaccharides, which are abundant, renewable biological macromolecules, have garnered considerable scientific interest due to their diverse pharmacological properties. Among these, β-glucans derived from the edible mushroom Lentinus edodes — commonly known as shiitake — have recently been illuminated for their compelling anticancer potential, particularly against cervical cancer cells. A groundbreaking study published in the high-impact journal Glycoscience & Therapy reveals an unprecedented molecular mechanism underlying the antitumor efficacy of Lentinus edodes-derived β-glucan (LNT), highlighting its direct interaction with a tumor suppressor protein and opening new paths for targeted cancer therapies.
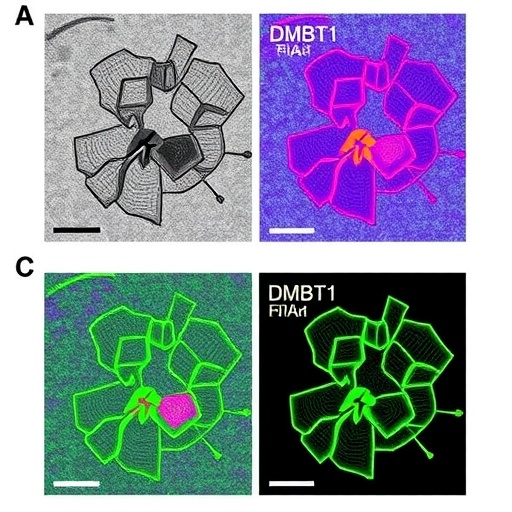
The researchers, led by a collaborative team from Wuhan University and the Shandong Laboratory of Yantai Drug Discovery in China, painstakingly decoded the biochemical crosstalk between LNT and human cervical cancer cells, specifically HeLa cells. Their findings pivot on the identification of DMBT1 (Deleted in Malignant Brain Tumors 1) as a crucial cellular target of LNT. This discovery represents the first time that DMBT1 has been implicated as a mediator for the anticancer activity of mushroom-derived β-glucans, setting a theoretical foundation for exploiting this axis in cervical cancer management.
At the heart of this molecular interaction is the binding affinity of LNT to DMBT1, a glycoprotein localized primarily on the cellular membrane of HeLa cells. The study elaborates on the formation of multiple non-covalent bonds — including hydrogen bonds, hydrophobic interactions, and van der Waals forces — that stabilize the LNT-DMBT1 complex. This specific binding not only fosters the upregulation of DMBT1 expression in a concentration-dependent manner but also triggers a cascade of intracellular events that suppress cancer cell proliferation and promote programmed cell death or apoptosis.
The intricate role of DMBT1 as a tumor suppressor was robustly validated using gene knockdown approaches. When DMBT1 expression was silenced in HeLa cells, the otherwise potent inhibitory effects of LNT on cancer cell growth were significantly mitigated. This attenuation establishes DMBT1 as an indispensable mediator in the antitumor activity of LNT. Intriguingly, the study also underscores the dampened modulation of the downstream PI3K/Akt signaling pathway in DMBT1-deficient cells, suggesting that LNT’s anticancer effects are closely tied to its ability to influence this critical oncogenic pathway via DMBT1.
PI3K/Akt, a prime signaling route that governs cell survival, growth, and metabolism, is frequently hyperactivated in cancer cells, fostering an environment conducive to uncontrolled proliferation and resistance to apoptosis. The revelation that LNT leverages DMBT1 to modulate this pathway introduces new mechanistic insights, positing that polysaccharide-based agents might offer targeted modulation of oncogenic signaling networks, circumventing the deleterious side effects often associated with conventional chemotherapy.
Beyond in vitro cell culture experiments, the team extended their exploration to in vivo models, substantiating the correlation between enhanced DMBT1 expression and the therapeutic efficacy of LNT. Tumor-bearing animal models demonstrated marked suppression of cervical cancer progression upon treatment with LNT, concurrent with upregulated DMBT1 levels. These findings corroborate the translational potential of LNT, heralding its promise as a biologically derived anticancer agent with a defined molecular target.
This multi-dimensional examination of LNT’s mechanism not only broadens the molecular understanding of how natural polysaccharides can be harnessed against cancer but also highlights the prospect of employing DMBT1 as a prognostic biomarker. The ability to stratify patients based on DMBT1 expression could enhance personalized treatment regimens, maximizing therapeutic success rates and minimizing unnecessary exposure to less effective drugs.
Moreover, the utilization of renewable natural products such as LNT aligns with the broader movement toward sustainable and less toxic treatment modalities. Compared to synthetic pharmaceuticals, β-glucans offer biocompatibility and potential synergistic effects when combined with existing chemotherapeutic agents, inviting further studies into combination therapies to combat cervical cancer more effectively.
Despite the promising findings, the study authors emphasize the necessity for further research to refine our understanding of LNT’s pharmacodynamics and pharmacokinetics in clinical contexts. Investigations into optimal dosing, delivery mechanisms, and potential systemic effects will be crucial steps toward integrating LNT into routine oncological practice.
This discovery also invigorates the field of glycoscience, an area investigating the role of carbohydrates in biological systems, by providing a tangible example of how polysaccharide-protein interactions can elicit profound biological outcomes. The intricate biophysical forces mediating the LNT-DMBT1 bond open avenues for designing synthetic analogs that may augment or mimic these natural interactions with enhanced specificity and efficiency.
In conclusion, the study elegantly bridges natural product chemistry and molecular oncology, presenting a compelling narrative of how a mushroom-derived β-glucan exerts its anticancer effects through a newly identified target, DMBT1. By extinguishing cervical cancer cell proliferation via modulation of the DMBT1–PI3K/Akt signaling axis, LNT emerges as a promising candidate for the development of novel, targeted therapies. This revelation not only advances therapeutic strategies but also underscores the vital role of natural compounds in the future landscape of cancer treatment.
The identification of DMBT1 as a pivotal target enriches the understanding of cervical carcinogenesis and offers strategic insights for therapeutic intervention. As cervical cancer remains a leading cause of cancer mortality among women globally, the development of safer and more efficacious treatments based on natural compounds is of paramount importance. The work of Hu, Li, and Xu thus represents a significant stride toward this critical goal, illuminating the path for further scientific inquiry and clinical innovation.
Subject of Research: Cells
Article Title: Lentinus edodes-derived β-glucan inhibits human cervical cancer progression through a potential target of DMBT1 on HeLa cell
Web References: http://dx.doi.org/10.1016/j.glycos.2026.100035
Image Credits: Shuqian Hu, Xuan Li, Xiaojuan Xu
Keywords: β-glucan, Lentinus edodes, cervical cancer, DMBT1, HeLa cells, PI3K/Akt pathway, apoptosis, tumor suppressor, polysaccharides, natural products, molecular oncology, targeted therapy
Tags: biologically active polysaccharides cancer treatmentDMBT1 mediatedDMBT1 tumor suppressor pathwayLentinus edodes β-glucan anticancer mechanismLentinus edodes β-glucan molecular mechanismmushroom-derived anticancer agentsnatural compounds in cancer therapyrenewable biological macromolecules anticancershiitake mushroom polysaccharides cervical cancertargeted cervical cancer therapiesβ-glucan HeLa cell interactionβ-glucan induced tumor suppression