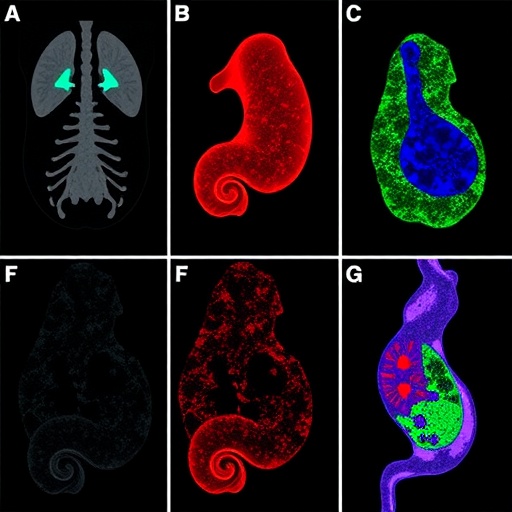
A groundbreaking breakthrough in cancer therapeutics has emerged from a collaboration between Johns Hopkins University and the University of Maryland School of Pharmacy, where researchers have engineered pioneering small-molecule drugs that simultaneously inhibit hypoxia-inducible factors 1 and 2 (HIF-1 and HIF-2). These transcription factors orchestrate a vast network of genes governing tumor survival, angiogenesis, and metastatic progression, particularly under hypoxic conditions commonly found within tumors. This novel dual inhibition strategy, published in the Journal of Experimental Medicine, heralds a new frontier in oncological treatment by seeking to incapacitate two master regulators of tumor biology in tandem.
HIF-1 and HIF-2 function as critical transcriptional activators that respond to low-oxygen (hypoxic) environments by modulating dozens of pathways crucial for tumor adaptation and progression. They drive angiogenesis—the formation of new blood vessels—to supply rapidly growing tumors with oxygen and nutrients, enhance tumor cell invasiveness, and subvert immune cell infiltration and function within the tumor microenvironment. The overexpression of these factors often portends poor prognosis and resistance to conventional therapies, highlighting the urgent need to develop therapeutics targeting these pathways.
Previously, selective inhibitors of HIF-2, such as belzutifan, have achieved clinical approval for cancers like advanced renal cell carcinoma. However, exclusive targeting of HIF-2 overlooks the complementary and distinct roles played by HIF-1 in cancer biology. This has propelled efforts to develop inhibitors with dual specificity, enabling a more comprehensive disruption of HIF-driven oncogenic processes. Researchers hypothesized that dual HIF-1/2 inhibition could overcome limitations of monotherapies and enhance therapeutic outcomes across a spectrum of tumor types.
At the heart of this drug discovery effort is an advanced computational drug design technology called Site-Identification by Ligand Competitive Saturation (SILCS). This method meticulously maps ligand-binding hotspots across protein surfaces based on structural data, enabling precise prediction of small molecules capable of binding target sites with high affinity. Using the known crystal structure of HIF-2, the team identified three conserved pockets shared between HIF-1 and HIF-2, thereby unlocking the potential for designing inhibitors active against both isoforms.
The researchers generated a focused library of candidate compounds predicted by SILCS to bind these conserved domains and subjected them to rigorous biochemical assays. Several molecules demonstrated robust binding to both HIF-1 and HIF-2, inducing proteasomal degradation of the transcription factors and effectively silencing downstream gene activation. These dual HIF inhibitors exhibited potent activity across multiple cancer cell lines in vitro, reducing expression of genes involved in angiogenesis, metabolism, and immune evasion.
Moving to in vivo models, the new drugs significantly impeded tumor growth in mouse models of breast, colorectal, melanoma, and prostate cancers. The inhibitors curtailed angiogenesis within tumors, diminishing blood vessel formation and thus nutrient supply, as well as reducing their propensity to invade adjacent tissue. Importantly, the treatment was well tolerated and administered orally, emphasizing clinical translatability.
Even more striking were the results when the dual inhibitors were administered alongside immune checkpoint blockade therapies such as anti-CTLA-4 or anti-PD1 antibodies. This combinatorial approach led to complete tumor remission in over half the treated animals, including those bearing tumors that were resistant to checkpoint inhibitors alone. Remarkably, these mice remained tumor-free long term and resisted rechallenge with fresh cancer cells, indicating a durable and systemic anti-tumor immune response had been established.
Detailed immunoprofiling revealed that dual HIF-1/2 inhibition reshaped the tumor immune microenvironment by reducing immunosuppressive cell populations, including myeloid-derived suppressor cells and regulatory T cells. Concurrently, there was an increase in cytotoxic T lymphocytes and natural killer (NK) cells, which are pivotal for cancer cell eradication, particularly when combined with checkpoint immunotherapy. This synergistic mechanism may explain the substantial efficacy of the combination regimen.
The discovery of these dual HIF inhibitors marks a significant conceptual advance, highlighting the therapeutic value of targeting conserved domains shared by paralog transcription factors involved in cancer progression. The translational potential is heightened by the oral bioavailability and tolerability observed in preclinical models. This approach could address unmet clinical needs related to hypoxic tumors, which are often refractory to conventional and immune-based therapies.
According to Gregg L. Semenza, a leading expert and co-senior author of the study, “The dual inhibition of HIF-1 and HIF-2 represents an exciting strategy for overcoming tumor hypoxia-induced resistance mechanisms, with the possibility to greatly expand the therapeutic landscape across multiple malignancies.” The fusion of computational design and experimental validation adopted in this study underscores the power of interdisciplinary efforts in accelerating drug development.
As cancer remains a multifaceted and heterogeneous disease, interventions that simultaneously disrupt multiple oncogenic pathways hold promise for improved patient outcomes. The dual HIF inhibitors exemplify such polypharmacology, capable of targeting the dynamic interplay between tumor metabolism, angiogenesis, and immune suppression. Future clinical trials will be essential to confirm efficacy and safety in humans, paving the way for a potentially paradigm-shifting class of anti-cancer therapeutics.
This innovative work also sets the stage for further exploration into combinatorial drug design targeting transcription factor families, which have traditionally been considered challenging constituents of the “undruggable” genome. The insights gleaned from targeting HIF-1/2 may inform strategies against other transcriptional regulators implicated in diverse diseases beyond oncology, expanding the horizons of precision medicine.
In summary, the development of small-molecule dual inhibitors against HIF-1 and HIF-2 transcription factors represents a transformative advance in cancer therapy. By leveraging state-of-the-art computational and experimental techniques, researchers have achieved comprehensive disruption of tumor adaptive pathways, demonstrating significant tumor regression and synergy with immunotherapy. These findings offer compelling hope for overcoming the longstanding clinical challenge posed by tumor hypoxia and advancing more effective, durable cancer treatments.
Subject of Research: Animals
Article Title: Targeting conserved domains of hypoxia-inducible factors for cancer therapy
News Publication Date: 2-Apr-2026
Web References: https://rupress.org/jem
References: Salman et al., Journal of Experimental Medicine, DOI: 10.1084/jem.20251009
Image Credits: © 2026 Salman et al., Originally published in Journal of Experimental Medicine
Keywords: Cancer, Breast cancer, Colorectal cancer, Skin cancer, Melanoma, Prostate cancer, Immunotherapy
Tags: breast cancer experimental treatmentcolorectal cancer mouse modelsdual HIF inhibition mechanismdual HIF-1 and HIF-2 small-molecule inhibitorshypoxia-inducible factors in cancer therapyimmunotherapy combined with HIF inhibitorsJohns Hopkins cancer research collaborationmelanoma tumor eradication strategiesovercoming tumor hypoxia resistanceprostate cancer novel therapeuticstargeting tumor microenvironment hypoxiatumor angiogenesis inhibition