In a groundbreaking development poised to reshape the landscape of immunology and drug discovery, researchers have unveiled a novel mechanism to directly modulate immune responses by targeting redox-sensitive cysteine residues on key immunological proteins. This study, spearheaded by Ng, M.Y., Nix, M.N., Du, G., and colleagues, marks a significant leap forward in understanding and manipulating macrophage inflammation, a central component of many diseases ranging from autoimmune disorders to chronic infections.
Immunological proteins are notoriously complex and multifaceted, with many serving as pivotal controllers of the body’s defense machinery. However, despite being heavily studied, a vast majority of these proteins have remained elusive to pharmacological intervention. This is largely due to challenges in identifying druggable sites that can reliably alter protein activity without detrimental side effects. The new research confronts this challenge head-on by exploiting the dynamic redox states of cysteine residues—amino acid components that can undergo oxidative modifications to act as molecular switches within cells.
Cysteine residues are particularly reactive due to their thiol side chains, capable of reversible oxidation and reduction, enabling them to function as signaling nodes within proteins. In immune cells, particularly macrophages, these redox modifications finely tune responses to pathogenic stimuli, such as bacterial lipopolysaccharides (LPS). The inability to systematically map these critical cysteine modifications across the immune proteome has hindered the exploitation of redox regulation for therapeutic purposes—until now.
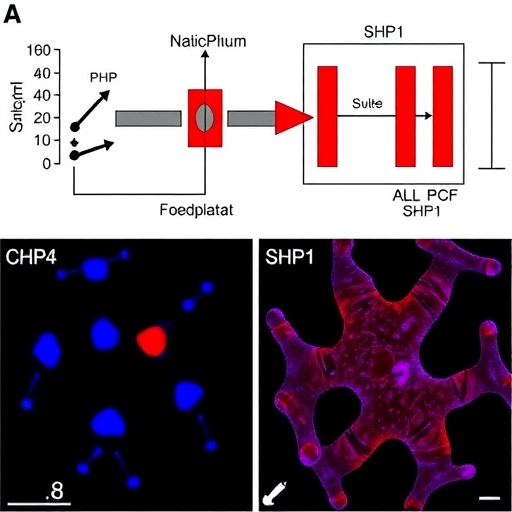
The researchers deployed an advanced deep redox proteomics approach to chart an unprecedented landscape of redox-sensitive cysteine sites in vivo. By analyzing proteins extracted directly from immune-relevant tissues, they identified a staggering 788 cysteine residues subject to redox regulation. These sites spanned a wide array of protein domains integral to immune function, offering a rich repository of potential regulatory hotspots previously unrecognized and untargeted by conventional drug design frameworks.
Among these findings, a particularly compelling discovery was the identification of a redox-sensitive cysteine at position 102 within the SHP1 protein—an immune regulator known for its critical role in modulating cytokine signaling pathways. SHP1 functions predominantly through an autoinhibitory mechanism, where its activity is cloaked by the N-SH2 domain until specific activation signals relieve this inhibition. The oxidation state of C102 emerged as a previously hidden switch that alters the conformation and activity of SHP1, thereby influencing immune responses.
Building on this insight, the team innovated a novel small molecule, termed SCA, designed to selectively and covalently bind to the cysteine residue at C102 within SHP1’s N-SH2 domain. This binding event effectively disengages the autoinhibition, activating SHP1’s phosphatase function. The targeted approach leverages the unique chemical reactivity of this redox switch, enabling precise modulation without broadly affecting other cysteines, thereby minimizing off-target effects.
Experimental validation in both mouse and human macrophages demonstrated robust engagement of SCA with SHP1 C102, confirming the compound’s specificity and mechanistic action. Functionally, this interaction antagonized the signaling pathways governed by interleukin-1 receptor-associated kinases (IRAKs), which play key roles in amplifying proinflammatory cytokine production in response to bacterial components like LPS. The net effect was a significant dampening of excessive macrophage-driven inflammation, highlighting SCA’s potential as an anti-inflammatory therapeutic agent.
Importantly, the therapeutic relevance of targeting SHP1’s redox switch extends beyond fine-tuning cytokine output. Macrophage inflammation is central to the pathogenesis of diverse conditions, including autoimmune diseases, sepsis, and metabolic syndrome. By introducing a chemically tractable switch that can dial down inflammatory signaling with high selectivity, this work lays foundational groundwork for a new class of immunomodulatory drugs that operate through redox regulation rather than classical receptor inhibition.
Beyond SHP1, the compendium of 788 redox-regulated cysteine sites represents a treasure trove for drug discovery. Each site embodies a potential regulatory node that can be harnessed to modulate immune function with tailored small molecules. This approach transcends traditional drug discovery paradigms that focus primarily on active sites or ligand-binding pockets, instead embracing the dynamic nature of post-translational modifications to unlock novel intervention strategies.
Moreover, the conceptual leap of exploiting redox biology as a therapeutic entry point resonates with a broader trend in chemical biology, where the intersection of proteomics, redox chemistry, and immune signaling converges to inform highly selective drug design. The methodology established by the authors provides a blueprint for future endeavors aimed at probing redox-regulated landscapes not only in immunity but in other physiological and pathological contexts.
The discovery of a druggable redox switch on SHP1 also illuminates broader biological principles. It underscores the sophisticated mechanisms evolved by immune cells to integrate metabolic and oxidative cues into functional outcomes. Understanding how redox modifications dictate protein conformation and signaling output will greatly enhance our grasp of immune cell plasticity and adaptability, laying the foundation for therapeutics that harmonize with intrinsic cellular regulatory networks.
This breakthrough may also inspire the design of covalent agonists that function via non-traditional mechanisms. While covalent inhibitors targeting cysteines are well-established, covalent agonists that activate rather than inhibit enzymes remain relatively rare. SCA exemplifies this strategy, binding irreversibly yet beneficially to a redox-sensitive cysteine to promote enzymatic activation, a paradigm that could be extended to other proteins with latent regulatory cysteines.
As the field moves forward, challenges such as achieving optimal tissue distribution, controlling off-target reactivity, and understanding long-term effects of redox modulation will need careful consideration. Nevertheless, this study highlights the enormous potential locked within redox-regulated proteomes and sets a high bar for future research and development.
In summary, the elucidation of a cysteine-based redox switch controlling SHP1 activity illuminates a new dimension of immune regulation and therapeutic opportunity. The advent of SCA as a selective covalent agonist heralds a new era where precise chemical control of immune cell signaling is achievable. This work not only advances our molecular understanding of macrophage biology but also charts a compelling course toward innovative redox-targeted immunopharmacology.
The implications of this discovery are wide-reaching, offering hope for better management of inflammatory diseases through novel molecular interventions. By bridging chemical biology with immunology, the study exemplifies how cutting-edge proteomic technologies combined with rational drug design can unlock next-generation therapies that were previously unimaginable.
As the scientific community digests these findings, excitement builds around the prospects of translating redox-targeted approaches into clinical interventions. The continued exploration of cysteine redox landscapes promises a renaissance in drug development, expanding the repertoire of actionable targets within the proteome and deepening our capacity to modulate human health at the molecular level.
Such integrative efforts will undoubtedly inspire innovation across disciplines, galvanizing a new wave of research dedicated to unveiling the therapeutic potential encoded within the redox chemistry of proteins—a chemical frontier with vast yet largely untapped promise.
Subject of Research:
Post-translational redox regulation of cysteine residues on immunological proteins and their pharmacological targeting to modulate macrophage inflammation.
Article Title:
A druggable redox switch on SHP1 controls macrophage inflammation.
Article References:
Ng, M.Y., Nix, M.N., Du, G. et al. A druggable redox switch on SHP1 controls macrophage inflammation. Nat Chem Biol (2026). https://doi.org/10.1038/s41589-026-02163-8
Image Credits:
AI Generated
DOI:
https://doi.org/10.1038/s41589-026-02163-8
Tags: cysteine oxidation in immune signalingdrug discovery for autoimmune inflammationimmune response regulation by redox switchesmacrophage inflammation modulationmolecular switches in immune cellsoxidative modifications in protein functionpharmacological targeting of immunological proteinsredox regulation of macrophage activityredox-based therapeutic strategiesredox-sensitive cysteine drug targetingSHP1 protein targeting in macrophagestargeting macrophage-driven chronic inflammation