In the complex landscape of hepatic surgery, post-hepatectomy liver failure (PHLF) continues to pose a significant clinical challenge, being one of the foremost causes of perioperative mortality in patients undergoing liver resection. Despite advances in surgical techniques and perioperative care, the early identification of patients at risk for clinically relevant post-hepatectomy liver failure (CR-PHLF) remains elusive. A new study from Zhejiang Provincial People’s Hospital, published in BMC Cancer, offers promising insights into predictive modeling that integrates biochemical and intraoperative parameters, potentially revolutionizing how surgeons anticipate and manage PHLF risk.
The research pivots on the understanding that timely detection of CR-PHLF is critical for patient outcomes, yet current predictive models leave much to be desired in accuracy and clinical usability. Traditional metrics, including the Albumin-Bilirubin (ALBI) score and Aspartate Aminotransferase to Platelet Ratio Index (APRI), provide some prognostic value but lack the sensitivity and specificity needed for confident clinical decision-making. Recognizing this gap, the research team conducted a retrospective analysis involving 915 patients who received liver resections at their facility, meticulously examining a wide range of variables to unearth more precise prognostic factors.
Central to this inquiry was the exploration of lactate as a biomarker. While lactate levels have historically been associated with tissue hypoxia and metabolic stress during surgery, their utility in predicting liver failure post-hepatectomy remains underexplored. By examining postoperative lactate in conjunction with demographic data, liver function tests, and intraoperative variables such as blood loss and transfusion requirements, the researchers aimed to develop a multifactorial predictive model that outperforms existing standards.
Multivariate logistic regression analyses identified six pivotal predictors that correlate strongly with the onset of CR-PHLF. These included gender, retention rate of indocyanine green at 15 minutes (ICGR-15), intraoperative blood loss, the necessity for transfusion, the extent of liver resection, and crucially, postoperative lactate levels. The inclusion of ICGR-15 underscores the importance of dynamic liver function assessment preoperatively, while intraoperative blood loss and transfusion reflect surgical stress and physiological derangement that may precipitate liver dysfunction.
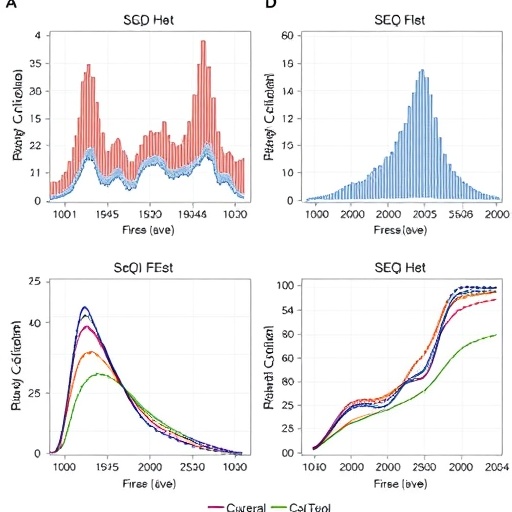
The standout feature of the developed model is its robust predictive accuracy, as evidenced by the area under the receiver operating characteristic curve (AUC). In the training cohort, the model achieved an AUC of 0.781—a marked improvement over traditional ALBI and APRI scores. Validation in an independent patient set yielded an even higher AUC of 0.812, suggesting strong reproducibility and potential for clinical implementation. This level of discriminative performance indicates that the model can reliably differentiate patients at high risk for developing clinically relevant PHLF from those with benign postoperative courses.
Underlying these statistical validations is the creation of a nomogram, a practical clinical tool translating complex mathematical risk assessments into intuitive charts. Such a nomogram facilitates bedside risk estimation by integrating individual patient data with the weighted contributions of identified predictors. By doing so, surgeons and perioperative teams can stratify patients pre- and intraoperatively, guiding more personalized decision-making regarding surgical extent, perioperative monitoring, and resource allocation.
The implications of this research extend beyond numerical improvements in predictive power. Incorporating lactate into a multivariate framework sheds light on the metabolic disturbances accompanying liver resection and their prognostic significance. Lactate accumulation post-surgery may reflect transient ischemia-reperfusion injury, impaired hepatic clearance, or systemic inflammatory responses — all pathophysiological mechanisms that can culminate in liver failure. By quantifying this biomarker alongside surgical and patient-specific factors, the model encapsulates a more holistic picture of perioperative risk.
Moreover, improvements in early detection of CR-PHLF can spearhead targeted interventions. For instance, high-risk patients identified through this model could benefit from enhanced hemodynamic support, vigilant ICU monitoring, or the early initiation of therapies like plasma exchange or liver-assist devices. Conversely, low-risk patients might avoid unnecessary resource-intensive treatments, optimizing healthcare efficiency.
This study notably emphasizes the heterogeneity of PHLF risk, highlighting how a constellation of factors rather than a single biomarker or clinical feature dictates outcomes. Gender differences identified as a predictor may reflect underlying variances in liver regenerative capacity or hormonal influences, while the extent of liver resection logically correlates with residual hepatic function. These nuances underscore the necessity of tailored rather than uniform perioperative management strategies.
It is worth acknowledging that while retrospective, this large-scale analysis offers a robust dataset that reflects real-world clinical populations, enhancing the external validity of the findings. However, future prospective studies and multi-center validations would be instrumental in confirming the model’s universal applicability across diverse patient demographics and surgical settings. Additionally, integrating emerging technologies such as machine learning could further refine predictive algorithms by discovering intricate nonlinear relationships among risk factors.
In an era where precision medicine increasingly informs surgical care, this integrated predictive model exemplifies the trajectory toward personalization of hepatic surgery. By marrying biochemical insights with intraoperative variables into a cohesive predictive framework, clinicians gain a powerful instrument to mitigate one of the most dreaded complications — posthepatectomy liver failure. The model’s superiority over existing tools not only represents a statistical triumph but offers tangible hope for improved perioperative outcomes and survival rates.
Beyond its clinical ramifications, this research also points toward the expanding role of lactate as more than a generic metabolic marker. As a dynamic, rapidly measurable parameter, lactate can potentially serve as an early warning signal flagging hepatic stress in the vulnerable postoperative period. This elevates its status from merely an observational variable to a strategic sentinel in perioperative monitoring protocols.
As liver surgeries grow in complexity and scope, incorporating extensive resections for malignancies and expanded donor criteria in transplant settings, precise risk stratification models are indispensable. The integration of this novel model into clinical pathways could transform preoperative planning, helping identify candidates who might benefit from neoadjuvant therapies, modified surgical approaches, or adjunctive support measures to improve hepatic resilience.
While the nomogram streamlines clinical workflow, the study’s findings also encourage multidisciplinary collaboration. Surgeons, anesthesiologists, hepatologists, and critical care specialists can leverage this risk stratification tool to orchestrate comprehensive care plans, emphasizing early detection, timely intervention, and prevention of liver failure sequelae. Ultimately, such synergy is crucial to translate predictive accuracy into improved patient prognoses.
In sum, this research heralds a leap forward in perioperative hepatology, combining metabolic biomarkers with surgical metrics to build a predictive model that significantly surpasses existing standards. Its capacity to anticipate clinically significant posthepatectomy liver failure promises to reshape both clinical practice and patient outcomes in liver surgery, addressing a pressing unmet need in the management of this life-threatening complication.
Subject of Research: Early detection and prediction of clinically relevant post-hepatectomy liver failure (CR-PHLF) using integrated predictive models combining lactate and intraoperative factors.
Article Title: Comparison of the accuracy of predictive models in early detection of clinically relevant posthepatectomy liver failure.
Article References:
Li, Y., Liu, YM., Gao, YL. et al. Comparison of the accuracy of predictive models in early detection of clinically relevant posthepatectomy liver failure. BMC Cancer 25, 1400 (2025). https://doi.org/10.1186/s12885-025-14738-0
Image Credits: Scienmag.com
DOI: https://doi.org/10.1186/s12885-025-14738-0
Tags: Albumin-Bilirubin score limitationsAspartate Aminotransferase Platelet Ratio Index analysisbiochemical parameters in liver surgeryearly identification of clinically relevant PHLFearly liver failure detectionimproving accuracy in liver failure predictionsinnovative approaches to managing liver failure risk.intraoperative parameters for liver failure predictionpost-hepatectomy liver failure risk assessmentpredictive modeling in liver surgeryprognostic factors for post-hepatectomy outcomesretrospective study on liver resection patients