In a groundbreaking study that could revolutionize the treatment approach for pediatric cancers, researchers have unveiled a novel therapeutic strategy targeting the proteostasis network in rhabdomyosarcoma (RMS), the most prevalent soft tissue cancer in children and adolescents. This research, published in the August 29, 2025, issue of Oncotarget, explores how interfering with cancer cells’ intrinsic protein quality control mechanisms can significantly hamper tumor growth, offering new hope for high-risk cases that poorly respond to conventional treatments.
Rhabdomyosarcoma is characterized by its aggressive nature and the challenge it presents in clinical treatment, especially in advanced or relapsed cases. Traditional modalities including chemotherapy and radiation therapy have demonstrated limited efficacy in eradicating the disease over the long term, prompting scientists to investigate alternate biological vulnerabilities within these malignancies. This study zeroes in on the proteostasis network—an intricate cellular system responsible for maintaining protein folding, trafficking, and degradation—which cancer cells exploit heavily to survive the heightened stress inflicted by rapid proliferation and genomic instability.
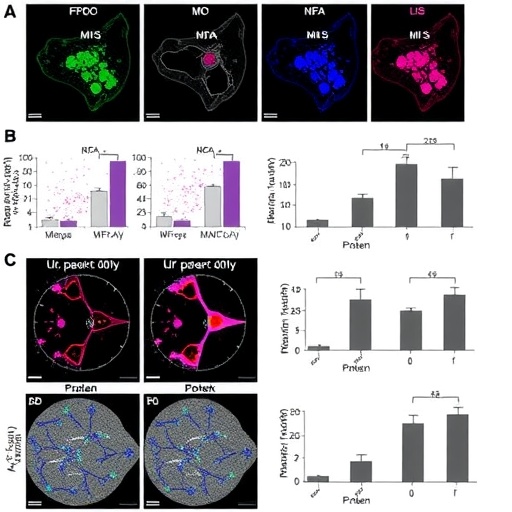
The investigative team, led by Kristen Kwong and Amit J. Sabnis at the University of California San Francisco’s Division of Pediatric Oncology, initially employed the compound MAL3-101 to disrupt proteostasis in RMS cells. MAL3-101, an inhibitor targeting the heat shock protein HSP70, impairs the chaperone machinery essential for protein homeostasis. Transcriptomic analyses of treated RMS13 cell lines revealed a suite of differentially expressed genes indicative of cellular stress and activation of the unfolded protein response (UPR), a conserved pathway that aims to restore protein folding capacity or initiate apoptosis when damage is irreparable.
Building upon these insights, the researchers harnessed computational tools such as SigCom LINCS to perform a systematic screen for genetic perturbations mimicking the transcriptomic signature induced by MAL3-101. This approach identified the loss of VCP—encoding the AAA ATPase p97—as a key node in the proteostasis network whose inhibition evokes similar stress phenotypes in various cancer cell lines. p97 coordinates several processes related to protein degradation and quality control, including endoplasmic reticulum-associated degradation (ERAD) and autophagy, making it a strategically compelling therapeutic target.
Pharmacological inhibition of p97 using potent compounds like CB-5083 and UPCDC-30766 in RMS models triggered a robust unfolded protein response characterized by PERK phosphorylation, splicing of XBP1 mRNA, and increased transcription of pro-apoptotic factors such as DDIT3. These molecular events culminate in cell death, delineating a mechanistic framework whereby proteostasis disruption compromises cancer cell viability. Notably, the treatment efficacy was demonstrated not only in vitro but also in vivo, where mouse xenograft models exhibited markedly reduced tumor progression upon administration of p97 inhibitors.
An intriguing facet of the study lies in the heterogeneous responses observed across different tumor specimens and cell lines. Some RMS models manifested resistance to p97 blockade through enhanced autophagic flux, a catabolic process enabling cells to recycle intracellular components and survive metabolic or proteotoxic stress. This adaptive mechanism appears to function as a compensatory survival pathway when the primary protein quality control network is compromised. Thus, autophagy activation emerges as a biomarker for resistance and a potential co-target in combinatorial strategies designed to augment therapeutic response.
The challenges posed by tumor heterogeneity and adaptive resistance underscore the complexity of targeting proteostasis in RMS. The investigators note that the genetic landscape of individual tumors profoundly influences their susceptibility to proteostasis inhibitors. These findings suggest a paradigm shift toward personalized medicine, wherein biomarkers of cellular stress pathways and autophagy are integrated into patient stratification to optimize treatment regimens. Furthermore, the combinational inhibition of compensatory pathways alongside p97 blockade could potentiate apoptosis and mitigate resistance.
This research not only delineates the molecular underpinnings linking proteostasis disruption to UPR activation and apoptosis but also propels the field toward novel drug development. While currently available p97 inhibitors demonstrate effectiveness, their clinical translation necessitates refinement for improved specificity and reduced off-target toxicity. The pursuit of safer, more drug-like compounds could translate into potent therapeutics that selectively dismantle cancer cell proteostasis without deleterious systemic effects.
The implications of this study extend far beyond rhabdomyosarcoma. Given the universal reliance of rapidly proliferating cancer cells on proteostasis networks to manage proteotoxic stress, similar strategies may prove efficacious against other tumor types notorious for therapeutic resistance. This avenue opens the door for a class of targeted treatments that fundamentally sabotage cancer cell survival strategies rather than solely aiming to kill cells with cytotoxic agents.
Notably, by targeting protein homeostasis pathways, scientists are beginning to exploit a vulnerability that is less prone to mutation-driven resistance mechanisms. Proteostasis is a highly conserved and essential process, and cancer’s heavy dependence thereupon could represent an Achilles’ heel. The capacity to induce irreversible cellular stress and trigger programmed death through UPR manipulation is both a promising and elegant therapeutic approach.
Looking ahead, clinical trials incorporating proteostasis inhibitors, alone or in combination with autophagy blockers and conventional therapies, will be essential to validate these preclinical findings in patient populations. Biomarker development for patient selection and response monitoring will also be critical components of future research efforts. Ultimately, this work sets the stage for a new era in pediatric oncology wherein molecularly informed, less toxic therapies can be tailored for children afflicted with aggressive cancers like rhabdomyosarcoma.
In summary, the manipulation of the proteostasis network via p97 inhibition represents a transformative strategy in targeting rhabdomyosarcoma. By dismantling cancer cells’ capacity to manage protein misfolding and stress, this approach leverages fundamental cellular processes to induce tumor regression. The study’s insights into resistance mechanisms and potential synergy with autophagy inhibitors underscore a sophisticated understanding of cancer biology that could reshape therapeutic paradigms and improve outcomes for some of the most vulnerable patients.
Subject of Research: Animals
Article Title: In vivo manipulation of the protein homeostasis network in rhabdomyosarcoma
News Publication Date: 29-Aug-2025
Web References:
http://dx.doi.org/10.18632/oncotarget.28764
Image Credits: © 2025 Kwong et al., distributed under CC BY 4.0
Keywords: cancer, protein homeostasis, rhabdomyosarcoma, unfolded protein response, preclinical therapeutics, p97
Tags: advanced cancer treatment approacheschallenges in treating rhabdomyosarcomachemotherapy limitations in pediatric cancerscombating soft tissue cancersinnovative therapies for high-risk cancersMAL3-101 inhibitor studypediatric cancer research advancementsprotein quality control in tumorsproteostasis network and cancerRhabdomyosarcoma treatment strategiestumor growth inhibition mechanismsUC San Francisco pediatric oncology research