NKCC1 is a human chloride transporter that has the ability to transport sodium, potassium, and chloride from the exterior into cells. In the kidney, for example, NKCC1 type proteins ensure that these ions are reabsorbed from the urine, and generally NKCC1 is important for osmotic cell volume regulation. In brain, NKCC1 and related proteins are important for chloride gradients that are vital for the electrical signaling in neuronal networks.
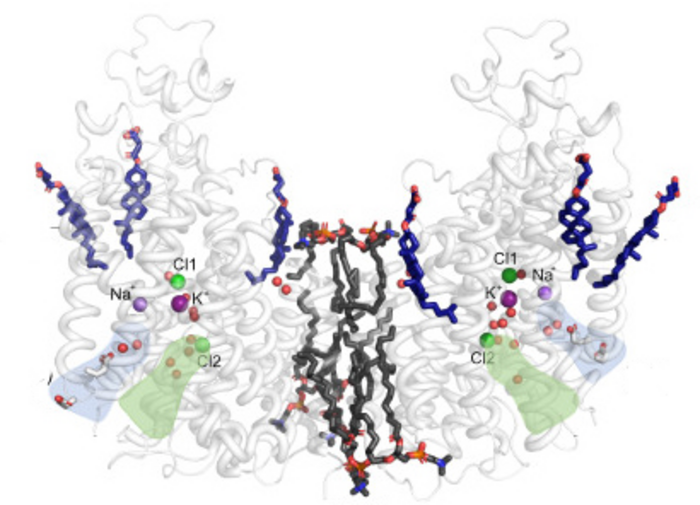
Credit: Caroline Neumann and Poul Nissen, Aarhus University
NKCC1 is a human chloride transporter that has the ability to transport sodium, potassium, and chloride from the exterior into cells. In the kidney, for example, NKCC1 type proteins ensure that these ions are reabsorbed from the urine, and generally NKCC1 is important for osmotic cell volume regulation. In brain, NKCC1 and related proteins are important for chloride gradients that are vital for the electrical signaling in neuronal networks.
Using cryo-electron microscopy (cryo-EM), a team from Poul Nissen’s laboratory with colleagues from the Fenton and Hartmann laboratories at Aarhus University (AU) and the Lindorff-Larsen laboratory at University of Copenhagen (KU) have determined the three-dimensional atomic structure of NKCC1 (a so-called Na+– K+– 2 Cl– cotransporter) and investigated its function.
Insights into the three-dimensional atomic structure and dynamics of NKCC1 including the bound ions, lipids and water molecules as well as ion transport studies in cells provide important new information on NKCC1 function, which is driven by the sodium gradient established by the sodium-potassium pump.
The studies that the team present in an article in EMBO Journal reveal a surprising mechanism for release of the ions into the cell that begins with one of the two bound chloride ions, and only then the sodium ion and finally the other chloride and the potassium ion. The researchers will now continue to try to identify novel compounds that interfere with NKCC1 function and that may help in for example kidney and brain disorders.
The project was highly challenging from the very beginning and was started almost 10 years ago as a collaboration between the Nissen and Fenton laboratories. First-author and PhD student Caroline Neumann (now graduated) and colleagues from the Nissen laboratory undertook important collaborations with Prof. Rune Hartman’s laboratory (AU) to establish an advanced expression system for efficient protein production, and with Prof. Robert Fenton’s laboratory (Dept. of Biomedicine, AU) for functional studies of the transporter in mammalian cells. Finally, a collaboration was initiated with Prof. Kresten Lindorff-Larsen’s group from the University of Copenhagen for the computational simulations of the NKCC1 dynamics and ion release.
Journal
The EMBO Journal
DOI
10.15252/embj.2021110169
Method of Research
Experimental study
Subject of Research
Cells
Article Title
Cryo-EM structure of the human NKCC1 transporter reveals mechanisms of ion coupling and specificity
Article Publication Date
14-Oct-2022