Sepsis is a high-mortality disease that arises when the body’s immune reaction to pathogens causes multi-organ defects. Although infection-induced pro-inflammatory cytokines are indispensable for pathogen elimination, dysregulated production of these factors can lead to endotoxic shock. Despite the extensive use of anti-TNF-α antibody administration or glucocorticoids in patients undergoing endotoxic shock, mortality rates remain high at 30%. These disappointing results suggest that the mechanism of endotoxic death is only partially explained by uncontrolled inflammation.
Studies of pain neurons have traditionally focused on the ion channels. Natural pain sensors such as the transient receptor potential cation channel subfamily V member 1 (TRPV1), and TRP ankyrin 1 (TRPA1) are expressed in afferent pain neurons and trigger pain to protect the organism from further harm. Emerging reports suggest that organ-innervated pain neurons and TRP channels also regulate dermal innate immunity, psoriasis, islet function, candidiasis, and osteomyelitis. Mechanistically, these phenomena are thought to be evoked by the paracrine secretion of nociceptor-derived peptides such as Calcitonin gene-related peptide (CGRP) and VIP, which modulate immune signals and endocrine pathways. These findings have also raised the question of whether pain neuron-derived “hormones” exist.
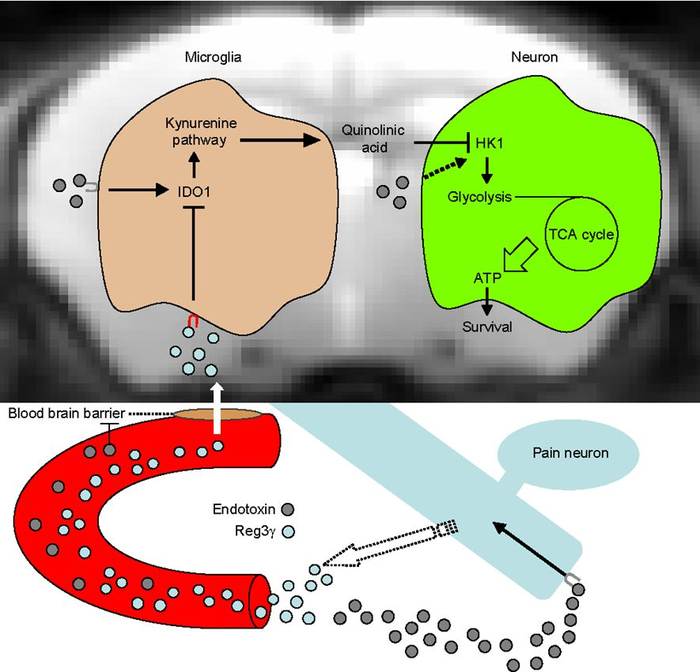
In this new study, Dr. Kenta Maruyama at NIPS, Assistant Professor Takeshi Kondo at Hokkaido University, and colleagues revealed that pain neuron-derived peptide Reg3γ penetrates the inflamed brain and suppresses the expression of microglial IDO1, a key enzyme of the kynurenine pathway. Endotoxin-administered pain neuron-null mice and pain neuron-specific Reg3γ deficient mice exhibit a high-mortality rate accompanied by decreased brain HK1 phosphorylation and ATP production despite normal inflammation. This metabolic arrest is only observed in the brain, and aberrant production of brain quinolinic acid, a neurotoxic metabolite of the kynurenine pathway, causes HK1 suppression. Notably, brain administration of Reg3γ protects mice from endotoxic death by enhancing brain ATP production. By identifying pain neuron-derived Reg3γ as a microglia-targeted hormone, this discovery provides novel insights into the understanding of tolerance to endotoxic death.
Credit: Kenta Maruyama
Sepsis is a high-mortality disease that arises when the body’s immune reaction to pathogens causes multi-organ defects. Although infection-induced pro-inflammatory cytokines are indispensable for pathogen elimination, dysregulated production of these factors can lead to endotoxic shock. Despite the extensive use of anti-TNF-α antibody administration or glucocorticoids in patients undergoing endotoxic shock, mortality rates remain high at 30%. These disappointing results suggest that the mechanism of endotoxic death is only partially explained by uncontrolled inflammation.
Studies of pain neurons have traditionally focused on the ion channels. Natural pain sensors such as the transient receptor potential cation channel subfamily V member 1 (TRPV1), and TRP ankyrin 1 (TRPA1) are expressed in afferent pain neurons and trigger pain to protect the organism from further harm. Emerging reports suggest that organ-innervated pain neurons and TRP channels also regulate dermal innate immunity, psoriasis, islet function, candidiasis, and osteomyelitis. Mechanistically, these phenomena are thought to be evoked by the paracrine secretion of nociceptor-derived peptides such as Calcitonin gene-related peptide (CGRP) and VIP, which modulate immune signals and endocrine pathways. These findings have also raised the question of whether pain neuron-derived “hormones” exist.
In this new study, Dr. Kenta Maruyama at NIPS, Assistant Professor Takeshi Kondo at Hokkaido University, and colleagues revealed that pain neuron-derived peptide Reg3γ penetrates the inflamed brain and suppresses the expression of microglial IDO1, a key enzyme of the kynurenine pathway. Endotoxin-administered pain neuron-null mice and pain neuron-specific Reg3γ deficient mice exhibit a high-mortality rate accompanied by decreased brain HK1 phosphorylation and ATP production despite normal inflammation. This metabolic arrest is only observed in the brain, and aberrant production of brain quinolinic acid, a neurotoxic metabolite of the kynurenine pathway, causes HK1 suppression. Notably, brain administration of Reg3γ protects mice from endotoxic death by enhancing brain ATP production. By identifying pain neuron-derived Reg3γ as a microglia-targeted hormone, this discovery provides novel insights into the understanding of tolerance to endotoxic death.
Journal
Cell Reports
Method of Research
Experimental study
Subject of Research
Animals
Article Title
Nociceptor-derived Reg3 prevents endotoxic death by targeting kynurenine pathway in microglia
Article Publication Date
8-Mar-2022