Credit: Yonatan Dubi & Yonatan Sivan
What happens to a piece of metal when you shine light on it? This question, which has been one of the driving forces of modern physics, gained renewed interest in recent years, with the advances in fabrication of small metallic nano-particles. When the piece of metal is very small, it turns out that it can couple extremely well to visible light. The study of fundamental and applicable aspects of this interaction is typically referred to as plasmonics.
Within the field of plasmonics – and considering metallic nanoparticles – two different answers emerged to the question posed above. The first, which relies on classical physics and is quite intuitive, is that the nanoparticle heats up. Indeed, the fact that illuminated nanoparticles serve of localized heat sources has found a wide variety of applications, from cancer treatment to water desalination. The second answer is gentler, and suggests that upon illumination, the electrons deviate from equilibrium and occupy a non-Fermi distribution, characterized by an excess of electrons at high energies, so-called “hot electrons”.
These two pictures, heating vs “hot electrons”, are typically presented as orthogonal, and theories either treat one or the other. In a recent work, conducted by the groups of Prof. Yonatan Sivan and Yonatan Dubi (both from Ben-Gurion University, Israel), these two pictures were merged into a single theoretical framework, which enabled them to fully evaluate both the electron distribution and the electron and lattice temperatures of an illuminated nanoparticle. Their research results were published in Light: Science and Applications.
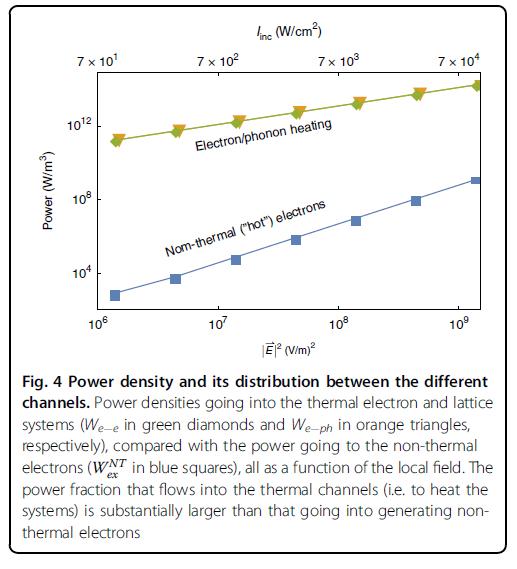
The picture that emerges from their study is that indeed the two effects – heating and generation of “hot electrons” – are present. Yet, in contrary to many recent claims, heating is far more important, and uses most of the illumination power input. Only a tiny fraction (less than one millionth) of the power input is channeled towards generation of “hot electrons”, which is thus an extremely inefficient process.
Many experimental and theoretical studies have celebrated the promise of exploiting “Hot Electrons” to perform various functions, from photo-detection to photo-catalysis. The work of Sivan and Dubi allows for a realistic evaluation of energy harvesting efficiency using “hot” electrons, and provides the limits of this efficiency. Furthermore, it serves as an essential first step towards realistic calculations of the complete energy harvesting process in many systems, from plasmonic-enhanced photo-catalytic systems to solar cells.
###
Media Contact
Yonatan Dubi
[email protected]
Related Journal Article
http://dx.




