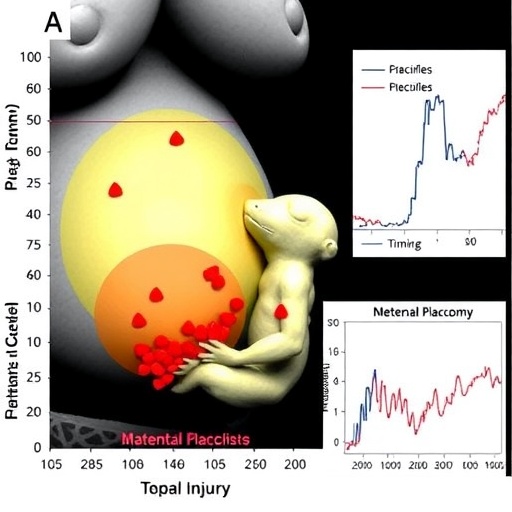
In a groundbreaking study that could revolutionize the diagnosis and management of neonatal encephalopathy (NE), researchers have delved into the intricate timing of hypoxic-ischemic injury, unveiling how these temporal dynamics influence long-term outcomes. Neonatal encephalopathy, a devastating disorder arising from insufficient oxygen and blood flow to the infant brain near the time of birth, remains a leading cause of neonatal mortality and lifelong neurological disability worldwide. Despite advances in neonatal critical care, predicting outcomes remains a complex clinical challenge, largely due to the multifaceted nature of brain injuries and their timing.
The research team employed highly sophisticated methods to dissect the temporal profiles of hypoxic-ischemic insult across the maternal-placental-fetal continuum. By defining precise timing patterns of oxygen deprivation events, the investigators sought to establish correlations between the duration and onset of hypoxic-ischemic episodes and the resulting neurological prognosis. This approach marks a significant shift from conventional models that often treat hypoxic-ischemic damage as a homogenous event occurring at or around birth.
Utilizing advanced biochemical and neuroimaging modalities, the researchers integrated data from maternal and placental biomarkers with neonatal clinical profiles to construct comprehensive temporal maps of injury. These maps reveal distinct phases of hypoxic-ischemic insult, highlighting critical windows during which damage accrual accelerates. Remarkably, the study elucidates that insults occurring in utero, several hours to days before delivery, engage different pathological trajectories compared to those commencing during labor or immediately postpartum.
One of the study’s pivotal contributions lies in demonstrating that the timing—not merely the severity—of the insult is intimately linked to the degree of neuronal loss, inflammation, and cerebral metabolic dysfunction. Earlier insults, potentially insidious and progressive, may allow partial compensatory mechanisms or subtle neuroplastic responses, whereas abrupt perinatal insults often trigger overwhelming cascade reactions leading to extensive cellular death. This nuanced understanding underscores the inadequacy of “one-size-fits-all” prognostic frameworks in neonatal neurocritical care.
The implications for clinical practice are profound. By stratifying neonates based on their defined temporal injury profiles, clinicians could tailor therapeutic interventions with greater precision. Early identification of prolonged in utero hypoxic events may prompt intensified antenatal monitoring or preemptive delivery strategies. Conversely, recognizing acute peripartum insults might prioritize urgent neuroprotective treatments immediately post-birth, including hypothermia or emerging pharmacotherapies targeting specific injury pathways.
Methodologically, the team deployed a multiparametric analytic framework combining high-resolution magnetic resonance imaging (MRI), electrophysiological monitoring, and molecular assays quantifying hypoxia-inducible factors (HIFs) and inflammatory mediators. This integrative model enabled cross-validation of injury timing derived from placental histopathology against in vivo neonatal brain signatures. The convergence of these modalities established temporal phenotypes with remarkable sensitivity and specificity.
Furthermore, longitudinal neurodevelopmental follow-ups at 6, 12, and 24 months provided robust outcome data correlating distinct hypoxic-ischemic timing profiles with cognitive, motor, and sensory deficits. Children whose injuries were characterized by antepartum hypoxia exhibited variable developmental delays but demonstrated better responses to early rehabilitative interventions, contrasting sharply with those suffering acute intrapartum insults who faced more severe and often irreversible impairments.
Ethical considerations also arise from the study’s findings. The ability to time hypoxic-ischemic injury with accuracy raises questions about prenatal counseling, perinatal decision-making, and long-term care planning. The research prompts a reevaluation of how information regarding injury timing should be communicated to parents, balancing hopeful prognostications with realistic expectations.
Moreover, the study catalyzes further inquiry into the molecular mechanisms underpinning differential responses to injury timing. For example, distinct gene expression patterns triggered by prolonged versus acute hypoxia may open avenues for targeted molecular therapies. Understanding how maternal-fetal interface dynamics influence vulnerability throughout gestation could illuminate preventative strategies.
The collaboration among neonatologists, obstetricians, neuropathologists, and biomedical engineers exemplifies the multidisciplinary approach necessary to tackle the complexity of neonatal brain injury. The innovative integration of biochemical assays with clinical neuroimaging sets a new standard for perinatal research methodology, offering replicable models adaptable to diverse healthcare settings.
Critically, this research addresses health disparities in neonatal outcomes by providing tools to detect subtle, previously elusive hypoxic-ischemic events. Early and accurate prognostication could reduce inequities by informing resource allocation and individualized care plans, especially in low-resource environments where standardized neuroimaging may be limited.
The findings ignite hope for the development of dynamic clinical protocols that incorporate real-time monitoring data to predict insult chronology. Future applications might extend beyond NE to other ischemic neonatal conditions, broadening the impact of this temporal profiling paradigm.
In conclusion, this landmark study redefines our understanding of hypoxic-ischemic insult timing in neonatal encephalopathy, establishing a crucial link between injury chronology and neurological prognosis. It heralds a new era in perinatal neuroscience, promising enhanced diagnostic accuracy, personalized therapeutic strategies, and ultimately improved outcomes for one of the most vulnerable patient populations—newborn infants grappling with the silent aftermath of oxygen deprivation.
Subject of Research: Hypoxic-ischemic insult timing profiles and prognosis in neonatal encephalopathy.
Article Title: Maternal-placental timing profiles of hypoxic-ischemic insult and association with prognosis in neonatal encephalopathy.
Article References:
Bills, H., Danguecan, A., Kinoshita, M. et al. Maternal-placental timing profiles of hypoxic-ischemic insult and association with prognosis in neonatal encephalopathy. J Perinatol (2026). https://doi.org/10.1038/s41372-026-02667-0
Image Credits: AI Generated
DOI: 13 April 2026
Tags: advanced neuroimaging in neonateshypoxic-ischemic injury timinghypoxic-ischemic insult phasesmaternal-fetal injury correlationmaternal-fetal oxygen deprivationmaternal-placental injury timingneonatal brain injury prognosisneonatal critical care advancementsneonatal encephalopathy diagnosisneonatal neurological outcomesplacental biomarkers in neonatestemporal dynamics of birth asphyxia