Spinal cord injuries (SCI) remain among the most daunting challenges in regenerative medicine and neurology, primarily due to their devastating and often lifelong consequences on motor and sensory function. The complexity of the spinal cord’s microenvironment following traumatic injury creates significant barriers to nerve fiber regeneration. One of the critical obstacles is scar tissue formation at the injury site, which obstructs the natural reparative processes and hinders functional recovery. Although the role of glial scars has been extensively analyzed, forming a partial barrier to axonal regrowth, the fibrotic component of scar tissue has historically been less thoroughly explored. This new pioneering study led by Prof. Xu Cao at Johns Hopkins University School of Medicine casts a spotlight on the molecular mechanisms underlying fibrotic scar formation, specifically the role of Transforming Growth Factor-beta 1 (TGF-β1), a cytokine intricately involved in fibrosis and tissue remodeling processes.
The investigation, published in the renowned journal Bone Research, presents compelling evidence linking TGF-β1 activity to fibrotic scarring and its resulting inhibitory effect on neural regeneration post-SCI. Prof. Cao and his team found that in adult murine models, the presence and induction of TGF-β1 at spinal injury sites initiate a cascade of cellular events that culminate in scar tissue development detrimental to neuronal recovery. Their groundbreaking work demonstrates that by suppressing the fibrotic response mediated by TGF-β1, significant improvements in motor and sensory function can be achieved, signifying a potential therapeutic avenue that could transform current treatment paradigms for SCI patients.
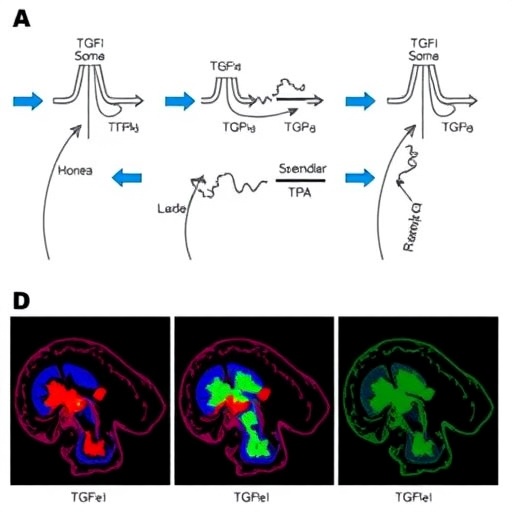
Utilizing an intricate animal model simulating human SCI, the researchers closely examined the cellular milieu at the lesion site. Among the pivotal findings was the identification of macrophages as a major source of activated TGF-β1 within the damaged spinal cord region. These macrophages, through autocrine and paracrine signaling, amplify the TGF-β1 pathway, triggering a recruitment of mesenchymal stromal stem cells (MSCs). These stem cells, upon entering the injury microenvironment, undergo differentiation into fibroblasts. Fibroblasts are the principal architects of fibrotic scar tissue, as they secrete extracellular matrix proteins such as collagen and fibronectin. The accumulation of such matrix molecules forms a dense and rigid scar, physically and biochemically obstructing axonal regeneration. Dr. Dayu Pan, the study’s first author, articulates that the overactivity of TGF-β1 orchestrates a deleterious feedback loop that perpetuates fibrotic scar formation via conversion of MSCs and local pericytes into pathogenic fibroblasts.
Intriguingly, the study elucidates that TGF-β1 signaling is not restricted to immune and progenitor cells but also impinges upon the vascular architecture of the spinal cord. Within the blood-spinal cord barrier niche, endothelial-associated pericytes respond to heightened TGF-β1 levels by transforming into fibroblasts themselves. This mechanism further potentiates the fibrotic matrix deposition at the injury site, compounding the barrier against axonal regrowth and functional repair. Prof. Cao highlights this novel cellular interaction as a critical insight, revealing how vascular-pericyte plasticity under TGF-β1 influence exacerbates scar density and complexity.
Recognizing the therapeutic potential of targeting this pathway, the research team employed selective inhibition of TGF-β1 within macrophage lineages. The resulting attenuation of TGF-β1 signaling led to a pronounced reduction in fibrotic scar tissue, allowing for enhanced penetration and preservation of nerve fibers within the lesion. Functional assessments via behavioral tests confirmed meaningful improvements in sensory and motor capabilities in mice subjected to this intervention. These findings underscore that macrophage-derived TGF-β1 is a principal driver of fibrosis-associated inhibition of spinal regeneration, presenting a tangible target for clinical treatment strategies.
Perhaps one of the most compelling revelations came from comparative analyses between adult and neonatal mice subjected to similar spinal cord injuries. Neonatal mice exhibited a striking absence of TGF-β1 activation at injury sites, correlating with the lack of fibrotic scar formation and seamless functional recovery. This stark contrast supports the hypothesis that aberrant activation of TGF-β1 in adulthood, as opposed to a more regulated or absent response in neonates, is a fundamental determinant in whether fibrotic scarring occurs and recovery is impeded. These developmental differences open new windows into understanding age-related disparities in regenerative capacity and scar pathology.
The study’s implications extend beyond merely understanding the biology of fibrosis in SCI. It advances the concept that modulating the immune response, specifically tuning macrophage activity and TGF-β1 signaling, can recalibrate the post-injury environment from one of inhibition to one supporting neural regeneration. Therapeutic work targeting this cytokine pathway, including neutralizing antibodies or receptor antagonists, has the potential not only to minimize scar formation but also to foster a more permissive milieu for axonal growth and functional restitution.
Beyond TGF-β1’s direct fibrotic roles, its intersection with vascular components implies that therapeutic approaches should consider the multifaceted cellular players mediating scar architecture—the interplay between immune cells, mesenchymal stem cells, pericytes, and fibroblasts. This study situates TGF-β1 as a master regulator of a complex cellular network dictating the spinal injury scar landscape and thereby influencing recovery outcomes.
Overall, the findings from Prof. Cao’s laboratory represent a quantum leap in spinal cord injury research and regeneration science. By illuminating the molecular culprits behind fibrotic scar formation, this work dismantles longstanding barriers to effective treatment development. The study offers a clear rationale for designing future therapeutics aimed at controlling excessive TGF-β1 activity and fibrotic scarring, which may profoundly improve quality of life for millions afflicted by SCI worldwide.
Future research trajectories could further explore the timing, dosage, and delivery mechanisms of anti-TGF-β1 therapies and their combinatorial use with other regenerative interventions, such as stem cell grafting or neurotrophic support. The work also encourages deeper investigation into neonatal spinal repair mechanisms, which may harbor key insights transferable to adult therapy development. As the field moves forward, integrating cellular and molecular insights like those provided here will be critical to overcoming the formidable challenge posed by SCI.
This landmark study, with robust experimental design, multidisciplinary approaches, and thoughtful emphasis on cellular crosstalk, sets a new research gold standard. It paves the way for translation from bench to bedside and ignites hope for a future when spinal cord injuries will no longer signify irreversible disability but rather an addressable clinical condition. The promise of harnessing the body’s own biology, modulated by precision molecular interventions, marks an exciting horizon in neuroscience and regenerative medicine.
Subject of Research: Animals
Article Title: TGF-β-induced fibrotic scar formation limits recovery of spinal cord injury
News Publication Date: 25-Feb-2026
References: DOI: 10.1038/s41413-026-00507-7
Image Credits: Dayu Pan from Johns Hopkins University School of Medicine
Keywords
Spinal cord injury, TGF-β1, fibrotic scar, nerve regeneration, macrophages, mesenchymal stromal stem cells, pericytes, fibrosis, neuroregeneration, extracellular matrix, blood-spinal cord barrier, functional recovery
Tags: animal models incytokines in tissue remodeling after SCIfibrotic scar formation in SCIglial versus fibrotic scar in SCIinhibitory effects of fibrotic scars on axonal regrowthJohns Hopkins spinal cord injury researchmolecular mechanisms of fibrosis in spinal injuryregenerative medicine for spinal cord traumaspinal cord injury recovery challengesTGF-beta 1 role in neural regenerationTGF-beta 1 signaling in spinal cord injurytransformative therapies targeting TGF-beta 1