In a groundbreaking study poised to redefine our understanding of fetal hematopoiesis, researchers have uncovered how mutations that reduce the expression of distinct ribosomal proteins—specifically RPS19 and RPL5—uniquely influence blood cell formation during development. Published in the prestigious journal Nature Communications, this work elucidates the complex regulatory roles of ribosomal subunits, challenging the conventional wisdom that ribosome dysfunction uniformly disrupts biological processes. The findings promise to open new avenues for targeted therapies in congenital anemias and related hematologic disorders.
Ribosomes, the cellular machines responsible for synthesizing proteins, have long been understood as essential but relatively uniform in their biological functions. Yet, emerging evidence suggests that ribosomes exhibit heterogeneity, with different ribosomal protein components imparting specific functional nuances. RPS19 and RPL5, two such ribosomal proteins, are notably implicated in Diamond-Blackfan anemia (DBA), a rare congenital disorder marked by defective red blood cell production. Until now, the mechanisms by which deficiencies in these proteins disrupt fetal hematopoiesis remained elusive.
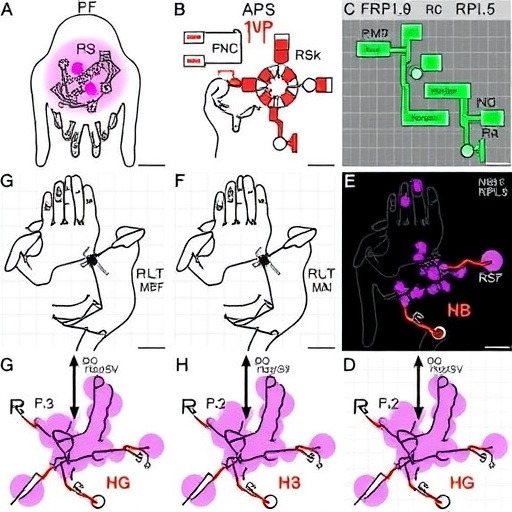
This study leverages sophisticated haploinsufficient models—genetically engineered to express reduced levels of either RPS19 or RPL5—to dissect their distinct contributions to fetal blood cell development. By comparing these models, the researchers uncovered a striking divergence in how the small and large ribosomal subunits influence hematopoietic stem cell differentiation and maturation. These differential effects underscore the previously underappreciated specialization of ribosomal proteins in orchestrating developmental processes.
The team applied state-of-the-art transcriptomic and proteomic analyses, unraveling the molecular pathways perturbed by each haploinsufficiency. Their data revealed that RPS19 deficiency primarily impairs early progenitor cell proliferation through disruptions in ribosomal RNA processing and altered protein synthesis fidelity. This results in a bottleneck at the earliest stages of erythroid lineage commitment, explaining the severe anemia observed in clinical DBA cases linked to RPS19 mutations.
Conversely, RPL5 haploinsufficiency manifests with a distinct cellular signature characterized by impaired maturation of erythrocytes and aberrant activation of stress response pathways. This suggests that large subunit deficiencies engender a qualitatively different type of ribosomal stress, one that influences cell fate decisions rather than proliferation per se. The nuanced regulatory differences identified reveal that ribosomal proteins operate far beyond mere structural roles within the ribosome complex.
Importantly, the findings challenge the oversimplified hypothesis that haploinsufficiency in ribosomal proteins uniformly triggers ribosomal stress responses and p53 activation. Instead, RPS19 and RPL5 reductions provoke distinct downstream signaling cascades, underscoring the need for tailored therapeutic strategies. For instance, interventions enhancing progenitor cell expansion may benefit RPS19-related disease, while modulating stress response pathways might better address RPL5-linked pathology.
Technically, the study’s use of CRISPR/Cas9 genome editing facilitated precise attenuation of gene expression levels, avoiding complete knockouts that often result in embryonic lethality. By preserving a haploinsufficient state, the models more faithfully recapitulate human disease phenotypes. This approach allowed for longitudinal studies of fetal hematopoietic development, revealing temporal windows when ribosomal protein function is critically required.
At a systems biology level, integrating multi-omics data unveiled a network of ribosome-associated proteins and signaling nodes differentially engaged by RPS19 and RPL5 deficiencies. Notably, the study identified novel interactors potentially mediating the divergent effects on hematopoietic progenitors. These interactors may serve as promising molecular targets to modulate ribosomal subunit-specific dysfunctions.
One of the most compelling aspects of the study lies in its potential translational impact. By distinguishing the discrete consequences of small versus large ribosomal subunit deficiencies, the research offers a conceptual framework for developing diagnostics that can stratify patients based on ribosomal protein mutation type. Such stratification could dramatically improve prognosis and enable precision medicine approaches to congenital anemias.
Moreover, these insights raise fundamental questions about ribosome heterogeneity in other developmental contexts and disease states. Could the differential roles of ribosomal proteins be harnessed to selectively control stem cell fate or combat malignancies characterized by ribosomal protein mutations? The study invites a broader reassessment of ribosome biology beyond its canonical functions.
The implications extend beyond hematopoiesis, as ribosomal dysfunction is increasingly recognized in a variety of genetic syndromes and cancers. This work provides critical evidence that the ribosome is not a monolithic entity but a dynamic participant in cell-specific regulatory networks. Decoding this complexity promises to unlock new therapeutic paradigms spanning regenerative medicine and oncology.
In summary, the innovative research by Tang, Ling, Mehmood, and colleagues compellingly demonstrates that RPS19 and RPL5 haploinsufficiency model systems reveal divergent controls by ribosomal subunits over fetal hematopoiesis. By dissecting the molecular underpinnings of these differences, the scientists have paved the way for future investigations into ribosome-mediated regulation of development and disease, spotlighting the ribosome as a sophisticated, multifaceted regulator of cell fate.
In the coming years, further exploration of ribosomal protein heterogeneity using advanced genetic models and high-resolution omics technologies will be essential for translating these foundational insights into clinical innovations. The paradigm shift heralded by this study underscores the urgency of redefining ribosome biology not merely as a cornerstone of protein synthesis but as an intricate node influencing the fate of life’s most essential cells.
As researchers continue to unravel the interplay between ribosomal subunits and cell lineage decisions, the prospects for targeted interventions in DBA and related disorders appear brighter than ever. This study not only deepens our molecular understanding but also exemplifies the power of precision genetic modeling in illuminating the subtleties of human developmental biology.
Subject of Research: Fetal hematopoiesis and the differential roles of ribosomal proteins RPS19 and RPL5 in blood cell development.
Article Title: RPS19 and RPL5 haploinsufficient models reveal divergent ribosomal subunit controls of fetal hematopoiesis.
Article References: Tang, Y., Ling, T., Mehmood, R. et al. RPS19 and RPL5 haploinsufficient models reveal divergent ribosomal subunit controls of fetal hematopoiesis. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71727-y
Image Credits: AI Generated
Tags: Diamond-Blackfan anemia ribosomal dysfunctiondivergent roles of ribosomal subunits in hematopoiesisfetal hematopoiesis regulation by ribosomal proteinsgenetic models of ribosomalribosomal protein mutations in congenital anemiaribosomal protein subunit-specific effectsribosome heterogeneity in blood disordersRPL5 haploinsufficiency effects on blood cell developmentRPS19 haploinsufficiency in fetal hematopoiesistargeted therapies for Diamond-Blackfan anemia