Lithium-ion batteries (LIBs) have become immensely popular as the go-to power source for a wide variety of electronic devices and vehicles over the past two decades. Although it is hard to overstate the transformative effects that LIBs have had on modern societies, this technology has a fair share of disadvantages that cannot be ignored any further. These include the limited availability of lithium as well as safety and environmental concerns. These drawbacks have motivated scientists around the world to look for alternative battery technologies, such as aqueous batteries. Potassium-ion batteries (KIBs) are a prominent example; these batteries are made from abundantly available materials and are much safer than LIBs. Moreover, KIBs can utilize a water-in-salt electrolyte (WISE), which makes them more stable thermally and chemically.
Credit: Shinichi Komaba from Tokyo University of Science
Lithium-ion batteries (LIBs) have become immensely popular as the go-to power source for a wide variety of electronic devices and vehicles over the past two decades. Although it is hard to overstate the transformative effects that LIBs have had on modern societies, this technology has a fair share of disadvantages that cannot be ignored any further. These include the limited availability of lithium as well as safety and environmental concerns. These drawbacks have motivated scientists around the world to look for alternative battery technologies, such as aqueous batteries. Potassium-ion batteries (KIBs) are a prominent example; these batteries are made from abundantly available materials and are much safer than LIBs. Moreover, KIBs can utilize a water-in-salt electrolyte (WISE), which makes them more stable thermally and chemically.
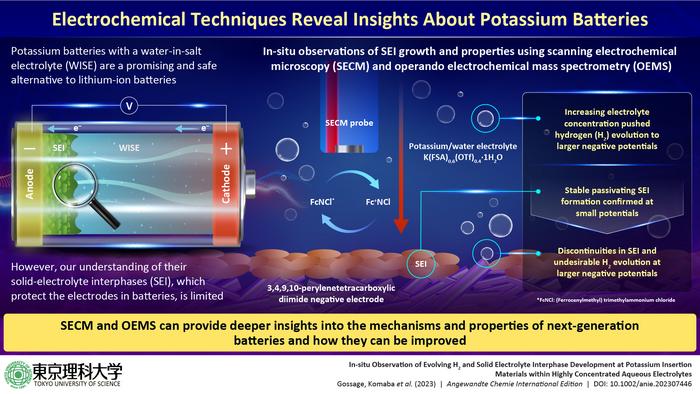
However, the prevention of hydrogen evolution at the negative electrode for its stabilization is a major challenge in high-voltage aqueous batteries. While solid-electrolyte interphases (SEI) that form between these electrodes and the electrolyte solution help stabilize the electrodes in LIBs (by preventing electrolyte decomposition and self-discharge of the batteries), they have been scarcely researched in the context of KIBs.
To address this major knowledge gap, a research team from Tokyo University of Science- (TUS), Japan, has recently conducted a pioneering study to gain insights into SEI formation and their properties in WISE-based KIBs. Their findings were published online in the journal Angewandte Chemie International Edition on August 18, 2023. The study, led by TUS Professor Shinichi Komaba, is co-authored by Junior Associate Professor Ryoichi Tatara, Dr. Zachary T. Gossage, and Ms. Nanako Ito, all from TUS.
The researchers mainly employed two advanced analytical techniques—scanning electrochemical microscopy (SECM) and operando electrochemical mass spectrometry (OEMS)—to observe how SEI forms and reacts in real time during the operation of a KIB with a 3,4,9,10-perylenetetracarboxylic diimide negative electrode and 55 mol/kg K(FSA)0.6(OTf)0.4∙1H2O, a WISE developed by the team in a previous study.
The experiments revealed that SEI forms a passivating layer in WISE akin to that seen in LIBs, with slow apparent electron transfer rates, helping suppress hydrogen evolution. This can ensure stable performance and higher durability of KIBs. However, the researchers observed that the coverage of the SEI layer was incomplete at higher operating voltages, leading to hydrogen evolution.
Taken together, the results reveal the need to explore potential avenues to enhance SEI formation in future aqueous batteries. “While our results reveal interesting details on the properties and stability of SEI found in one particular WISE, we should also focus on reinforcing the SEI network to achieve improved functionality,” comments Prof. Komaba. “SEI could perhaps be improved by the development of other electrolytes that produce unique SEIs, but also through the incorporation of electrolyte additives or electrode surface pretreatment.”
This study also highlights the power of SECM and OEMS for gaining a solid understanding of electrode–electrolyte interactions in next-generation batteries. “These techniques provide a powerful means for tracking the development, coverage, ion transfer, and stability of SEI and can easily be adapted for a variety of electrolytes and electrodes,” explains Prof. Komaba. “We hope that this work encourages other researchers to further explore SECM and OEMS as advanced characterization methods that can be incorporated with traditional battery measurements to gain deeper insights.”
The development of aqueous batteries such as KIBs will be instrumental for sustainable societies in the future, since they could replace the expensive and hazardous LIBs currently used in electric vehicles, smart grids, renewable energy systems, and marine applications. By making energy storage more accessible, aqueous batteries will aid the transition toward carbon-neutral energy generation, paving the way for a greener future.
With any luck, further studies will lead us to promising LIB alternatives soon!
***
Reference
DOI: https://doi.org/10.1002/anie.202307446
About The Tokyo University of Science
Tokyo University of Science (TUS) is a well-known and respected university, and the largest science-specialized private research university in Japan, with four campuses in central Tokyo and its suburbs and in Hokkaido. Established in 1881, the university has continually contributed to Japan’s development in science through inculcating the love for science in researchers, technicians, and educators.
With a mission of “Creating science and technology for the harmonious development of nature, human beings, and society”, TUS has undertaken a wide range of research from basic to applied science. TUS has embraced a multidisciplinary approach to research and undertaken intensive study in some of today’s most vital fields. TUS is a meritocracy where the best in science is recognized and nurtured. It is the only private university in Japan that has produced a Nobel Prize winner and the only private university in Asia to produce Nobel Prize winners within the natural sciences field.
Website: https://www.tus.ac.jp/en/mediarelations/
About Professor Shinichi Komaba from Tokyo University of Science
Prof. Shinichi Komaba obtained his Ph.D. from Waseda University, Japan, and then joined Iwate University as a research associate. After studying as a post-doctoral researcher at Bordeaux-CNRS, France, he joined Tokyo University of Science in 2005 to work on developing electrodes, electrolytes, and binding materials for various types of rechargeable batteries. His research group carries out cutting-edge research in the field of rechargeable batteries and their electrochemical applications. With more than 290 publications to his credit, Prof. Komaba has won numerous international awards, including the title of “Highly Cited Researcher” from 2019 to 2022.
https://www.tus.ac.jp/en/fac/p/index.php?486f
About Junior Associate Professor Ryoichi Tatara from Tokyo University of Science
Dr. Ryoichi Tatara graduated from Yokohama National University in 2012 and obtained a Ph.D. degree there in 2017. He joined Tokyo University of Science as an Assistant Professor in 2020 and was later promoted to Junior Associate Professor. His research focuses on energy-related chemistry, including electrodes, electrolytes, binders, and additives for lithium-, sodium-, and potassium-ion batteries. He has published around 100 research papers, which have been cited over 2,000 times.
Funding information
This study was partially funded by the JST-CREST (Grant No. JPMJCR21O6), MEXT Program: Data Creation and Utilization Type Materials Research and Development Project (JPMXP1121467561), and JSPS KAKENHI (Grant No. JP20H02849). Ryoichi Tatara thanks the TEPCO memorial foundation research grant (basic research).
Journal
Angewandte Chemie International Edition
DOI
10.1002/anie.202307446
Method of Research
Experimental study
Subject of Research
Not applicable
Article Title
In-situ Observation of Evolving H2 and Solid Electrolyte Interphase Development at Potassium Insertion Materials within Highly Concentrated Aqueous Electrolytes
Article Publication Date
18-Aug-2023