Antithrombotic therapy is prescribed to prevent thrombotic (blood clotting inside a blood vessel) events but the treatment also increases the likelihood of experiencing a bleeding event, which can be extremely serious if it occurs in a vital organ. Ageing societies tend to have an increased number of patients undergoing antithrombotic therapy, and the drugs used in this treatment can affect kidney function. In particular, reduced kidney function caused by antithrombotic medications can significantly influence bleeding events. It is highly recommended that patients, especially those with decreased kidney function, have a detailed discussion with their doctor about the possible risks and benefits of proceeding with antithrombotic therapy.
Credit: Kunihiko Matsui, MD, MPH of the Department of General Medicine and Primary Care, Kumamoto University Hospital, Kumamoto, Japan
Antithrombotic therapy is prescribed to prevent thrombotic (blood clotting inside a blood vessel) events but the treatment also increases the likelihood of experiencing a bleeding event, which can be extremely serious if it occurs in a vital organ. Ageing societies tend to have an increased number of patients undergoing antithrombotic therapy, and the drugs used in this treatment can affect kidney function. In particular, reduced kidney function caused by antithrombotic medications can significantly influence bleeding events. It is highly recommended that patients, especially those with decreased kidney function, have a detailed discussion with their doctor about the possible risks and benefits of proceeding with antithrombotic therapy.
Patients with heart arrythmia (atrial fibrillation) have a high risk for thrombotic events in blood vessels that could lead to permanent organ damage—such as cerebral infarction—and are prescribed antithrombotic therapy to lower their risk of developing blood clots. However, the risk of bleeding events simultaneously increases due to the nature of these medications. The severity of these bleeding events is highly variable, ranging from epistaxis (nosebleeds) to fatal brain hemorrhage.
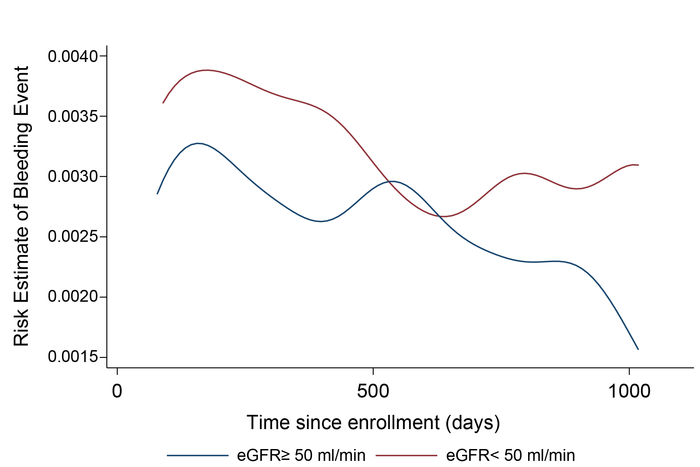
While kidney function is known to be related to bleeding event risk, researchers at Kumamoto, Miyazaki, and Tohoku Universities in Japan conducted a post-hoc subgroup analysis of the Atrial Fibrillation and Ischemic Events with Rivaroxaban in Patients with Stable Coronary Artery Disease (AFIRE) trial to determine the impact of kidney function on the risk of recurrent bleeding events during antithrombotic therapy. Their analysis revealed that the effect of kidney function on recurrent bleeding risk events was quite large for patients undergoing this treatment. They also found that the bleeding risk decreased with time for patients with healthy kidney function but remained high for patients with decreased kidney function. Clearly, the decision to use such a therapy should be balanced between the expected antithrombotic effects and bleeding risks.
In most cases, it is considered better to continue antithrombotic therapies even after bleeding events as long as the event was not severe. However, it is not surprising that both patients and physicians hesitate to continue the therapy after any bleeding event. To assess for drug safety and efficacy, these drugs are usually measured by the numbers of bleeding and thrombotic events. Unfortunately, in the assessment of antithrombotic therapy, most studies only consider the first event in their analyses even though patients could experience multiple events throughout their lifetime. This study revealed that the impact of kidney function on bleeding risk during antithrombotic therapy is larger than estimated in previous studies. Furthermore, patients with healthy kidney function appear to have a decreased risk of experiencing a bleeding event over time, but the risk for patients with reduced kidney function remains high as time continues.
“A detailed discussion between patients and physicians based on all current scientific evidence about the risks and benefits of antithrombotic therapy is highly recommended,” said study leader Dr. Kunihiko Matsui of Kumamoto University Hospital’s Department of General Medicine and Primary Care. “Our analysis should be quite useful in facilitating this type of discussion.”
Journal
BMC Medicine
DOI
10.1186/s12916-022-02268-6
Method of Research
Randomized controlled/clinical trial
Subject of Research
People
Article Title
The impact of kidney function in patients on antithrombotic therapy: a post hoc subgroup analysis focusing on recurrent bleeding events from the AFIRE trial
Article Publication Date
25-Feb-2022
COI Statement
KMa has no relationships relevant to the contents of this paper to disclose. SY reports grants from Takeda Pharmaceutical, Abbott, and Boston Scientific and personal fees from Daiichi-Sankyo and Bristol-Meyers. KKa reports grants from Grants-in-Aid for Scientific Research (20K08451) from the Ministry of Education, Culture, Sports, Science and Technology of Japan and grants and personal fees from Bayer Yakuhin and Daiichi Sankyo. MA reports grants from the Japan Agency for Medical Research and Development (AMED), personal fees from Bristol-Myers Squibb and Nippon Boehringer Ingelheim, and grants and personal fees from Bayer Yakuhin and Daiichi Sankyo. JA reports personal fees from Bayer Yakuhin and Sanofi and grants and personal fees from Daiichi Sankyo. TM reports grants from Japan Cardiovascular Research Foundation and personal fees from Nippon Boehringer Ingelheim, Daiichi Sankyo, Astra Zeneca, and Bayer Yakuhin. MN reports grants and personal fees from Bayer Yakuhin, Daiichi Sankyo, and Sanofi and personal fees from Bristol-Myers Squibb and Nippon Boehringer Ingelheim. KMi reports personal fees from Amgen Astellas BioPharma, Astellas Pharma, MSD, Bayer Yakuhin, Sanofi, Takeda Pharmaceutical, Daiichi-Sankyo, Nippon Boehringer Ingelheim, and Bristol-Myers Squibb. NH reports grants and personal fees from Bayer Yakuhin, grants from Nippon Boehringer Ingelheim, and personal fees from Bristol-Myers Squibb. KKi reports grants from the Japan Cardiovascular Research Foundation; grants and personal fees from Bayer Yakuhin, Daiichi Sankyo, Sanofi, MSD, and AstraZeneca; and personal fees from Bristol-Myers Squibb and Nippon Boehringer Ingelheim. AH reports grants and personal fees from Boston Scientific Japan, Otsuka Pharmaceutical, Sanofi, Astellas Pharma, Bristol-Myers Squibb, Daiichi Sankyo, and Bayer Yakuhin; grants from Fukuda Denshi, Abbott Japan, Japan Lifeline, Takeda Pharmaceutical, and Sumitomo Dainippon Pharma; and personal fees from Toa Eiyo, Nippon Boehringer Ingelheim, Amgen Astellas BioPharma, and AstraZeneca. HO reports personal fees from Towa Pharmaceutical, Bristol-Meyers Squibb, Pfizer, Toa Eiyo, Bayer Yakuhin, and Novartis Pharma.