In a groundbreaking study published in Nature Chemical Biology, researchers have unveiled the critical role of tau phosphorylation in the functionality of protective tau envelopes. This research sheds light on the intricate mechanisms underlying tau protein behavior, particularly in the context of neurodegenerative diseases such as Alzheimer’s. The implications of these findings are vast, as they provide new insights into how cellular processes can be disrupted by post-translational modifications, specifically phosphorylation, which is a significant contributor to the pathology of tau-related disorders.
The tau protein has garnered increasing attention due to its association with neurodegenerative diseases. Recognized for its role in stabilizing microtubules in neuronal cells, tau’s improper phosphorylation is known to lead to the formation of neurofibrillary tangles, one of the hallmark features of Alzheimer’s disease. Understanding the regulatory mechanisms of tau phosphorylation is essential for developing targeted therapies aimed at alleviating the symptoms and progression of such diseases.
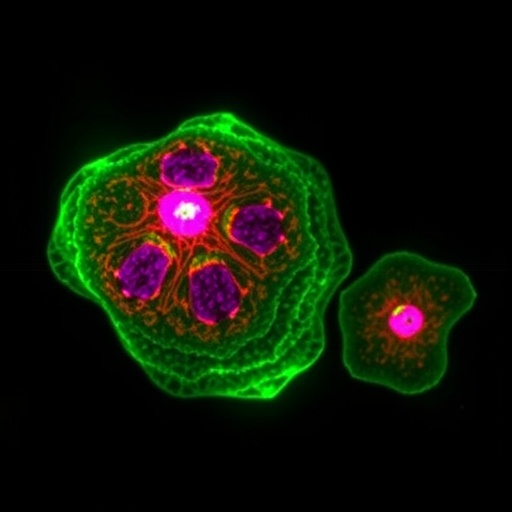
The focus of the study conducted by Siahaan and colleagues delves deep into the relationship between tau phosphorylation and its protective envelopes. These envelopes, formed by tau proteins, serve crucial functions in cellular defense, particularly under stress conditions. The research team employed advanced biochemical techniques to elucidate how phosphorylation alters the structural conformation of tau and, consequently, its ability to form and maintain these protective structures.
Utilizing a combination of in vitro assays and cellular models, the researchers meticulously characterized the effects of specific phosphorylation sites on tau. Their findings indicate that hyperphosphorylation, which typically occurs in pathological conditions, significantly impairs the ability of tau to aggregate into these protective envelopes. This impairment raises important questions regarding how tau’s functionality is compromised in diseased states and emphasizes the need for further exploration into therapeutic strategies that target tau modifications.
In a series of experiments, the team demonstrated that when tau is phosphorylated at critical serine and threonine residues, its capacity to interact with microtubules and maintain structural integrity is drastically reduced. The altered binding dynamics under these conditions suggest that phosphorylated tau not only loses its stabilizing effects on microtubules but also becomes toxic to neuronal cells. The dual roles of tau—both protective and detrimental—reveal the complexity of its function in the brain.
The implications of impaired tau envelope functionality extend beyond just structural roles. The study highlights how these envelopes play a part in cellular signaling pathways that are vital for neuronal survival. When tau phosphorylation disrupts this signaling, it can precipitate a cascade of events that lead to cell death, a defining characteristic of neurodegenerative diseases. Consequently, restoring the balance of tau phosphorylation could represent a promising therapeutic avenue.
Siahaan and his team speculate that their findings may also apply to other tauopathies, illnesses characterized by similar neurodegenerative processes due to tau dysfunction. The need for a nuanced understanding of tau’s behavior through the lens of post-translational modifications is paramount as researchers and clinicians alike seek to unravel the complexities of these debilitating diseases.
In the grander context of neurobiology, this research contributes to an evolving narrative about the interplay between protein modification and cellular health. The dynamics of phosphorylation not only influence tau proteins but potentially extend to a myriad of other proteins implicated in various cellular processes. This broad spectrum highlights a critical area for future neurobiological research—understanding how post-translational modifications can serve as modifiable risk factors for neurodegeneration.
As the scientific community digests these findings, the potential for targeted interventions focused on tau phosphorylation opens new doors for treating age-related cognitive decline and neurodegeneration. Advances such as small molecule inhibitors or monoclonal antibodies aimed at specific phosphorylation sites may represent practical approaches in clinical settings, allowing for more tailored therapeutic strategies.
While the pathway to clinical application is still fraught with challenges, the results of this study provide a robust framework for future research. As an intriguing prospect, the ability to engineer tau proteins that resist phosphorylation or to enhance the expression of phosphatases responsible for dephosphorylating tau could yield groundbreaking advancements in treatment modalities.
Moreover, the authors encourage a multidisciplinary approach to tackle this intricate issue, combining efforts from structural biochemistry, molecular biology, and clinical neuroscience. Implementing a collaborative framework will undoubtedly accelerate the development of clinical solutions that align closely with the underlying mechanisms of tau pathologies.
The study thus stands as not only a significant contribution to the understanding of tau biology but also as a call to arms for researchers, clinicians, and pharmaceutical companies alike to actively engage in the quest for effective treatments that could one day alter the course of neurodegenerative diseases. With this newfound knowledge, the scientific community takes a powerful step forward in addressing one of the most pressing health challenges of our time.
As we move forward into an era of precision medicine, the insights gained from the study of tau phosphorylation and its effects on protective tau envelopes may lead to innovative therapeutic strategies and ultimately reshape the landscape of neurodegenerative disease treatment.
By unravelling the complexities of tau phosphorylation, we stand on the cusp of potential breakthroughs that could significantly enhance our understanding and management of neurodegenerative disorders, paving the way for improved quality of life for countless individuals around the globe. The journey from basic research to clinical application may be long, but with studies like these lighting the way, hope is on the horizon.
Subject of Research: Effects of tau phosphorylation on protective tau envelopes and its implications for neurodegenerative diseases.
Article Title: Tau phosphorylation impedes functionality of protective tau envelopes.
Article References:
Siahaan, V., Weissova, R., Karhanova, A. et al. Tau phosphorylation impedes functionality of protective tau envelopes.
Nat Chem Biol (2026). https://doi.org/10.1038/s41589-025-02122-9
Image Credits: AI Generated
DOI: https://doi.org/10.1038/s41589-025-02122-9
Keywords: tau phosphorylation, neurodegenerative diseases, Alzheimer’s, protective envelopes, protein modification, therapeutic strategies
Tags: Alzheimer’s disease pathologybiochemical techniques in neurosciencecellular defense mechanisms in neuronsimplications of tau phosphorylationneurodegenerative diseases researchneurofibrillary tangles formationpost-translational modifications in tauprotective tau envelopes functionalitytargeted therapies for tau-related disorderstau envelopes structural integritytau protein behavior studytau protein phosphorylation