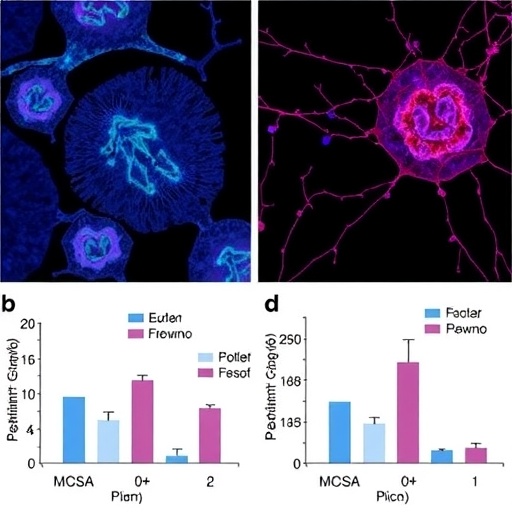
In a groundbreaking study set to redefine our understanding of tumor immunology and metabolic signaling, researchers Liu, Geng, Huang, and colleagues have unveiled a striking connection between the pentose phosphate pathway (PPP) and the cGAS-STING signaling axis. Their findings, published in Nature Communications in 2026, illuminate how metabolic reprogramming within the tumor microenvironment modulates the function of intratumoral conventional dendritic cells (cDCs), thereby enhancing antitumor immunity. This revelation opens promising avenues for therapies targeting metabolic and immune pathways to potentiate cancer immunotherapy.
Dendritic cells serve as pivotal sentinels of the immune system, orchestrating immune responses by processing and presenting tumor antigens to T cells. However, within the suppressive tumor microenvironment, their function often becomes impaired, limiting effective immune surveillance and tumor eradication. The study explores the metabolic underpinnings that restore and augment dendritic cell efficacy, focusing on the interaction between the PPP—a central metabolic route involved in nucleotide biosynthesis and redox homeostasis—and the innate immune sensor pathway cGAS-STING, known for detecting cytosolic DNA and triggering type I interferon production.
The pentose phosphate pathway, traditionally viewed through the lens of its biosynthetic and antioxidant roles, is now implicated as a crucial amplifier of immune signaling mechanisms. The researchers meticulously demonstrate that enhanced flux through the PPP bolsters cGAS-STING activation, culminating in heightened dendritic cell immunogenicity. This metabolic-immune crosstalk empowers cDCs within tumors to overcome immunosuppression, thereby promoting robust T cell-mediated antitumor responses.
At the core of their mechanistic dissection, Liu et al. reveal that metabolites generated via the PPP serve not merely as building blocks or redox buffers but also as signaling mediators that amplify the cGAS-STING pathway. For instance, the availability of ribose sugars and nicotinamide adenine dinucleotide phosphate (NADPH) was shown to be essential for the optimal activation of cGAS, the cytosolic DNA sensor, and its downstream effector, STING. This finding disrupts the traditional compartmentalization of metabolic and immune pathways, highlighting an integrated cellular strategy that leverages metabolism to boost innate immune surveillance.
Moreover, the study observes that the functional enhancement of cDCs by the PPP-cGAS-STING axis catalyzes a cascade of immunostimulatory events within the tumor microenvironment. These include elevated production of type I interferons and pro-inflammatory cytokines, critical for the recruitment and activation of cytotoxic T lymphocytes. Such metabolic conditioning of dendritic cells enriches the immunogenic landscape of tumors, potentially transforming “cold,” immunologically inert tumors into “hot,” immune-active sites susceptible to immunotherapy.
This work also addresses the spatial and temporal dynamics of metabolic flux within tumor-resident dendritic cells. Using advanced imaging and metabolomic profiling, the authors map increases in PPP activity coinciding with cGAS-STING pathway engagement during dendritic cell priming. This correlation underscores a deliberate cellular adaptation that coordinates metabolism with innate immune sensing to optimize antitumoral defense.
Importantly, the research team demonstrates that pharmacological or genetic modulation of key PPP enzymes directly influences dendritic cell activation and tumor control efficacy. In mouse tumor models, enhancing PPP activity correlated with improved antigen presentation and T cell activation, leading to significant tumor regression. Conversely, inhibition of PPP components impaired cGAS-STING signaling and blunted antitumor immunity, confirming the causal role of this metabolic pathway in immune function.
Beyond dendritic cells, the implications of this discovery ripple through the broader fields of cancer biology and immunometabolism. The data suggest that targeting the PPP might synergize with existing immune checkpoint blockade therapies, potentiating durable antitumor immunity. It also provides a rationale for developing therapeutic agents aimed at modulating metabolic flux to fine-tune immune responsiveness selectively within tumors, minimizing systemic toxicity.
Furthermore, this study challenges prevailing paradigms that view metabolic pathways predominantly as tumor-promoting mechanisms due to their role in sustaining cancer cell proliferation. Instead, it highlights their dualistic nature, serving as vital players in immune activation and tumor suppression depending on cellular context and pathway compartmentalization. Such insights encourage a reappraisal of metabolic interventions in oncology for more nuanced, context-specific therapeutic strategies.
On the molecular front, the investigation brings to light how the redox state maintained by PPP-derived NADPH is indispensable for the stability and activity of cGAS-STING signaling complexes. This NADPH provision shields signaling components from oxidative inactivation, ensuring sustained pathway engagement and robust downstream gene expression. Hence, metabolic health of dendritic cells emerges as a fundamental determinant of innate immune competence within tumors.
The team also explores how DNA damage and cellular stress within dendritic cells induce a feedback loop that further stimulates PPP activity, creating a self-reinforcing circuit that magnifies immune signaling. This feedback mechanism may act as a metabolic timer or amplifier for dendritic cell activation following antigen uptake, aligning metabolic support with functional demand.
Parallel investigations within this study analyze the transcriptomic shifts accompanying PPP-driven cGAS-STING activation, identifying gene expression programs that favor dendritic cell maturation, migration, and cytokine production. These transcriptional landscapes provide molecular markers for functional dendritic cell states linked to metabolic enhancements, offering new biomarkers for monitoring immunometabolic therapies.
As the tumor microenvironment is a complex arena of immunosuppressive signals, nutrient competition, and hypoxia, the discovery that metabolic reprogramming through the PPP selectively enhances dendritic cell function is especially meaningful. It implies that even in nutrient-deprived or hostile environments, strategic metabolic adaptations can resurrect effective immune surveillance and break tumor tolerance.
Looking ahead, this pioneering work sets the stage for translational research aimed at harnessing the PPP-cGAS-STING axis for clinical benefit. Therapeutic strategies could involve metabolic adjuvants that increase PPP flux specifically in dendritic cells or combined regimens with STING agonists to amplify antitumor immunity synergistically. The precise targeting of metabolic pathways within immune cells offers a tantalizing prospect for increasing the precision and efficacy of cancer immunotherapies.
Ultimately, the study by Liu and colleagues not only enriches the fundamental scientific understanding of immune metabolism but also heralds a new frontier in cancer therapy. By revealing how a vital metabolic pathway fuels innate immune signaling to empower dendritic cells, it bridges two previously siloed disciplines, opening pathways toward innovative, metabolism-based immunomodulation strategies to combat cancer more effectively.
The interplay between metabolic reprogramming and immune activation in tumor-resident dendritic cells highlighted here underscores the intricacy of cellular adaptation within the tumor microenvironment. This insight prompts reevaluation of immune cell function from a strictly immunologic perspective towards a more integrated metabolic-immune framework, potentially revolutionizing therapeutic approaches in oncology and immunotherapy.
In sum, the PPP’s fueling of cGAS-STING signaling within intratumoral conventional dendritic cells emerges as a critical nexus for sustaining robust antitumor immunity. Such metabolic-immune crosstalk not only enhances our biological understanding but also presents actionable targets for developing next-generation immune interventions, heralding a new era of metabolism-driven cancer immunotherapy.
Subject of Research: The integration of the pentose phosphate pathway metabolism with cGAS-STING innate immune signaling in enhancing the function of intratumoral conventional dendritic cells.
Article Title: Pentose phosphate pathway fuels cGAS-STING signalling to boost function of intratumoral conventional dendritic cells.
Article References: Liu, B., Geng, Z., Huang, Y. et al. Pentose phosphate pathway fuels cGAS-STING signalling to boost function of intratumoral conventional dendritic cells. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70934-x
Image Credits: AI Generated
Tags: antitumor immunity enhancement strategiescancer immunotherapy targeting metabolismcGAS-STING signaling in cancerintratumoral conventional dendritic cells functionmetabolic modulation of immune responsesmetabolic reprogramming of dendritic cellsmetabolic-immune pathway interactions in tumorsnucleotide biosynthesis in immune cellspentose phosphate pathway in tumor immunologyredox homeostasis in tumor immunitytumor microenvironment and immune suppressiontype I interferon production in cancer