In the realm of medical research, the establishment of reliable models to study complex disease states is paramount. Recent advancements have led to innovative methodologies, including the development of patient-derived organoids. These three-dimensional structures mimic the physiological architecture and functionality of organs. They serve as a valuable platform for understanding disease mechanisms, screening drugs, and investigating pathophysiological states. A recent study conducted by Zhang et al. touches upon a groundbreaking application of organoids in the study of primary aldosteronism, a condition characterized by excessive secretion of the hormone aldosterone, causing secondary hypertension and a variety of cardiovascular complications.
Primary aldosteronism, often underdiagnosed, significantly contributes to cases of secondary hypertension that are resistant to conventional therapy. Research has shown that the prevalence of primary aldosteronism is higher than previously acknowledged, impacting a sizable portion of the population. In this context, the establishment of organoids derived from the affected patients presents a novel avenue to explore the hormonal and cellular dysregulations inherent in this disorder. Zhang and colleagues have constructed a xenograft model that integrates patient-derived organoids with advanced imaging technologies to bridge the gap between preclinical research and clinical application.
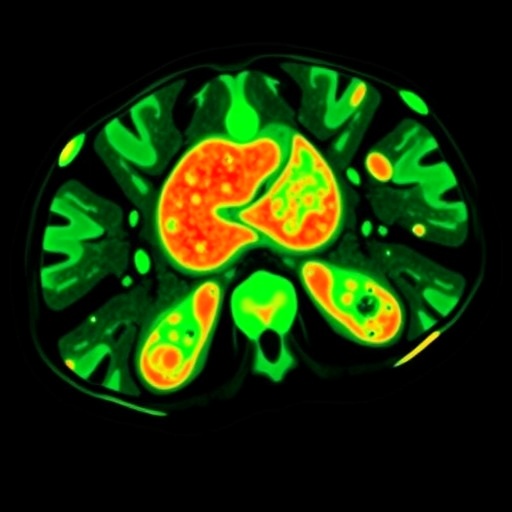
The methodology employed in this study not only highlights the utility of organoids in modeling disease but also integrates advanced imaging techniques to offer a comprehensive view of the disease mechanisms. The researchers used [^18F]AlF-NOTA-pentixather, a radiotracer known for its selective binding properties, in combination with positron emission tomography/computed tomography (PET/CT) to visualize the dynamics of aldosterone secretion in these organoid models. This combination establishes a robust platform for real-time imaging, allowing for a deeper understanding of the implications of excessive aldosterone levels in a genetically and biochemically relevant setting.
The study’s findings indicate that these organoids retain the phenotypic characteristics of the original tissue from which they were derived. This fidelity is crucial, as it enables researchers to observe how aldosterone affects cellular behaviors and tissue integrity in a controlled environment reminiscent of actual human physiology. Furthermore, the imaging capabilities offered by PET/CT allow scientists to monitor the biodistribution and metabolic activity of the organoids over time, effectively creating a bridge between bench-side research and the clinical cohort.
One of the critical aspects addressed in the research is the adaptability of the organoids for various therapeutic tests. The combination of organoids with imaging technology offers a dynamic platform for drug screening that can directly correlate treatment responses with physiological changes. This means that new therapeutic agents can be tested in real-time, significantly accelerating the drug development pipeline and providing more precise information on their efficacy before moving into clinical trials.
Moreover, this technique allows for the identification of potential biomarkers associated with treatment response. As the organoids react to therapeutic interventions, they can provide data on which molecular pathways are activated or inhibited. This information can be invaluable in personalizing treatment strategies for patients suffering from primary aldosteronism and potentially other related endocrine disorders. The implications are profound, as personalized medicine continues to gain traction in contemporary healthcare, focusing on tailoring treatments to individual biological profiles rather than a one-size-fits-all approach.
Further exploration into the different cellular interactions within the organoids reveals complexities that cannot be captured by traditional two-dimensional cell culture systems. The three-dimensional architecture allows for unique cell-cell interactions and microenvironmental factors that influence cell behavior, drug metabolism, and response to treatment. Such nuances can only be appreciated through the lens of advanced imaging and organoid technology.
The study highlights that ongoing research is essential for refining this model. While the early results are promising, further validation against a broader array of patient samples and larger cohorts is necessary. This will aid in confirming the reproducibility and scalability of the organoid model as a platform for studying primary aldosteronism and other related disorders. Moreover, integrating multi-omics approaches could enhance our understanding of the biological systems at play.
The implications of these findings extend beyond mere academic interest. As healthcare systems increasingly focus on chronic disease management, understanding the underlying mechanisms in conditions like primary aldosteronism is crucial. This model not only provides insights into disease pathophysiology but also lays the groundwork for actionable strategies that can significantly improve patient outcomes. Clinicians could apply this knowledge to refine diagnostic protocols, identify at-risk populations, and implement targeted preventative measures against complications associated with primary aldosteronism.
In conclusion, the establishment of a patient-derived organoids-based xenograft model coupled with imaging technologies represents a significant step forward in the study of primary aldosteronism. As researchers continue to unveil the intricate relationship between aldosterone and its physiological effects through advanced methodologies, we can anticipate a shift in how we approach diagnosis, treatment, and understanding of hormone-related diseases. This research not only opens the door to potentially revolutionary therapeutic strategies but also emphasizes the importance of innovative models in bridging the gaps between preclinical and clinical research.
With ongoing research in the field, it is likely that the insights gained from this study will catalyze a broader movement towards personalized therapeutics in endocrinology. As technologies evolve and more intricate relationships between hormones, tissues, and diseases become elucidated, the future of medicine may very well lie in the further integration of organoid technology and real-time imaging.
Subject of Research: Patient-derived organoids in primary aldosteronism research.
Article Title: Establishment and evaluation of a patient-derived organoids-based xenograft model of primary aldosteronism using [18F]AlF-NOTA-pentixather PET/CT: bridging preclinical and clinical imaging.
Article References: Zhang, X., Mou, T., Wang, Y. et al. Establishment and evaluation of a patient-derived organoids-based xenograft model of primary aldosteronism using [18F]AlF-NOTA-pentixather PET/CT: bridging preclinical and clinical imaging. J Transl Med 23, 927 (2025). https://doi.org/10.1186/s12967-025-06979-1
Image Credits: AI Generated
DOI: 10.1186/s12967-025-06979-1
Keywords: Primary aldosteronism, patient-derived organoids, xenograft model, PET/CT, imaging technology, personalized medicine, drug screening, disease mechanisms.
Tags: advancements in organoid technology for clinical researchcardiovascular complications of primary aldosteronismhormonal dysregulation in hypertensionimaging technologies in disease studyinnovative methodologies in organoid researchnovel organoid model for primary aldosteronismorganoids for drug screening and disease mechanismspatient-derived organoids in medical researchpreclinical research applications of organoidssecondary hypertension and aldosteroneunderdiagnosed conditions in hypertensionxenograft models in disease modeling