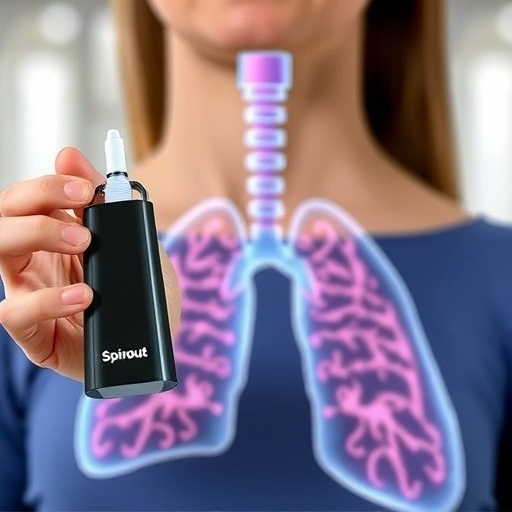
In a groundbreaking advancement for respiratory medicine, PulmoBioMed Ltd, a distinguished spinout from Northumbria University, has secured CE marking for its innovative PBM-Hale™ breath sampling platform. This regulatory milestone signifies the first-ever approval for a device capable of reliably collecting saliva-free exhaled breath condensates (EBC) from the deep lung, a feat that has eluded medical technology for over four decades. This innovative non-invasive device promises to revolutionize lung disease diagnostics by providing access to critical respiratory biomarkers without the need for invasive procedures.
The PBM-Hale™ platform represents a monumental shift in how clinicians approach lung disease sampling. Traditionally, deep lung samples have been obtained via invasive methods such as bronchoscopy and bronchoalveolar lavage. These procedures, while effective, pose significant risks to patients and involve complex, costly instrumentation that is not feasible for routine or large-scale use. Most patients can only be assessed through symptomatic treatment due to these limitations, hindering the accurate diagnosis of underlying pulmonary conditions. PBM-Hale™ overcomes these barriers by enabling direct breath condensate collection from the alveolar regions, offering clinicians a non-invasive, safe, and repeatable diagnostic tool.
This device’s ability to isolate breath condensates from the deep lung while excluding contamination from the mouth, upper airways, and ambient environment is a critical innovation. Such contamination has long compromised the specificity and reliability of breath-based diagnostics. PulmoBioMed’s technology uses a patented condensation and filtration system, developed through rigorous peer-reviewed research, to capture pure samples representative of the pulmonary microenvironment. This ensures that clinicians and researchers receive uncontaminated data, enhancing the diagnostic accuracy for a range of lung pathologies such as chronic obstructive pulmonary disease (COPD), asthma, interstitial lung disease, and even lung transplant monitoring.
The pathway to this achievement began within the research laboratories of Northumbria University, where Professor Sterghios Moschos pioneered the design and proof-of-concept studies underlying PBM-Hale™. Under his guidance, extensive clinical trials validated the device’s sampling precision and ease of use across diverse healthcare settings. Funding from Northern Accelerator, a collaborative commercialization partnership among northeastern UK universities, accelerated development from initial concept to functional prototypes. This effort also benefited from rapid prototyping techniques, including 3D printing within Northumbria’s engineering facilities, marking a seamless transition from academia to commercialization.
Professor Moschos, serving as both Founder and CEO of PulmoBioMed, underscores the impact of this technology: “We have addressed a respiratory diagnostic challenge dormant for four decades by enabling non-invasive access to deep-lung samples. Now with CE marking, PBM-Hale™ can integrate into clinical workflows and catalyze personalized respiratory medicine.” His vision emphasizes the device’s role in enabling precise, actionable data to guide therapeutic decision-making and accelerate drug development pipelines in pulmonology.
The manufacturing scalability and quality assurance of PBM-Hale™ have been entrusted to Sanner GmbH, a USFDA-registered Contract Development and Manufacturing Organization (CDMO) based in Germany. Sanner’s advanced expertise expedited the device’s evolution from prototype to a market-ready product, ensuring stringent compliance with medical device regulatory frameworks. Stefan Verheyden, CEO of Sanner, lauded the collaboration, emphasizing how PulmoBioMed’s innovative approach aligns with a broader medical movement toward safer, more accessible respiratory diagnostics.
PBM-Hale™’s introduction to clinical and research environments is poised to impact multiple facets of respiratory medicine. For lung transplant centers, it offers non-invasive, repeatable monitoring options critical for early detection of rejection or infection. Pharmaceutical developers stand to benefit from PBM-Hale™’s ability to provide deep-lung biomarkers that can refine clinical trial endpoints and enhance the assessment of novel respiratory therapeutics. The device’s seamless compatibility with existing diagnostic workflows further facilitates its adoption in routine practice and research settings.
The technology also has profound implications for patient safety and comfort. By eliminating invasive sampling, PBM-Hale™ minimizes procedure-related risks such as bleeding, infection, and anesthesia complications. This democratizes respiratory diagnostics, enabling broader screening and longitudinal monitoring programs in both hospital and community healthcare systems. Ultimately, this eases the burden on healthcare infrastructure while empowering personalized medical strategies tailored to individual pulmonary conditions.
The CE marking granted to PulmoBioMed’s device is also a reflection of meticulous scientific validation. The platform was subjected to randomized controlled clinical trials that demonstrated its reliability in capturing uncontaminated samples indicative of the deep lung environment. These trials substantiated its diagnostic value and reinforced the device’s role as a key player in the evolving landscape of breath-based medicine, significantly improving upon prior generations of breath analysis technologies.
In addition to the clinical advantages, PBM-Hale™ sets a new standard for interdisciplinary innovation. The confluence of biomedical engineering, pulmonology, and advanced manufacturing techniques exemplifies how collaborative research ecosystems can accelerate the translation of scientific breakthroughs into real-world healthcare solutions. PulmoBioMed’s journey from university lab to market-ready device showcases how sustained investment in translational research and commercialization partnerships can yield transformative medical technologies.
The successful introduction of PBM-Hale™ into European and American markets heralds a new era in respiratory diagnostics, with broader implications for global health. As respiratory diseases remain among the leading causes of morbidity and mortality worldwide, enhanced diagnostic tools capable of early detection and personalized treatment stratification could dramatically improve patient outcomes. PulmoBioMed’s device stands at the forefront of this paradigm shift, embodying the future of non-invasive pulmonary medicine.
Looking forward, PulmoBioMed is actively establishing structured collaborations with lung transplant centers, diagnostic manufacturers, research institutions, and pharmaceutical companies to expand PBM-Hale™’s clinical and research applications. Early initiatives are focused on environments where consistent and reliable access to deep-lung samples can transform clinical decision-making and therapeutic development. These efforts underscore the device’s potential to catalyze advances across diverse sectors of respiratory healthcare.
As the demand for precise, minimally invasive diagnostic platforms escalates, PulmoBioMed’s PBM-Hale™ exemplifies innovation that responds directly to clinical needs while maintaining rigorous scientific and regulatory standards. This first-of-its-kind CE-marked device opens promising new avenues for understanding pulmonary diseases, tailoring interventions, and ultimately improving respiratory health worldwide.
Subject of Research: People
Article Title: CE-Marked Breath Sampling Device PBM-Hale™ Enables Non-Invasive Deep Lung Diagnostics
News Publication Date: Not specified
Web References:
– https://pulmobiomed.com/
– https://northernaccelerator.org/
References: Clinical trial data and peer-reviewed research underpinning PBM-Hale™ (specific citations not provided)
Image Credits: PulmoBioMed
Keywords
Medical Device, Lung Disease, Respiratory Medicine, Breath Sampling, Exhaled Breath Condensate, Non-Invasive Diagnostics, Pulmonary Biomarkers, Personalized Medicine, CE Marking, Northumbria University, PulmoBioMed, Deep Lung Sampling
Tags: alternatives to bronchoscopy and lavageCE marked breath sampling technologyclinical respiratory biomarker extractiondeep lung breath condensate collectionnon-invasive lung disease diagnosticsNorthumbria University spinout medical devicePBM-Hale breath sampling platformpeer-reviewed breath analysis validationpulmonary biomarker detection technologyrespiratory medicine innovationsafe lung disease sampling methodssaliva-free exhaled breath condensate device