Credit: Edward Partlow and Dr. Richard Baker
Endocytosis, a fundamental process that cells use to take in macromolecules, functions a lot like an airlock on a spaceship – but squishier, says Dr. Gunther Hollopeter, assistant professor of molecular medicine at the Cornell University College of Veterinary Medicine. Researchers have studied how cells initiate and perform endocytosis since the 1960s, but in a new paper in the journal eLIFE, Hollopeter’s lab finally describes how cells shut down this important cellular machinery. And their findings are not without controversy.
“Until our work, people had not appreciated that it’s important to turn this process off,” said Hollopeter. Endocytosis functions in neurodegenerative diseases like Alzheimer’s, viral infections, cancer and cardiovascular disease. “That’s why it’s important for us to understand how this machinery is working. If we could figure out how to turn it up or down, and tune it in different organ systems, maybe we could control some of these disease progressions.”
Endocytosis is crucial for everyday functioning of all types of cells. “The type of endocytosis we study is a multipurpose tool the cells use – it’s very adaptable,” said Edward Partlow, a graduate student in the Hollopeter lab. For example, the process helps determine how cells respond to signals that tell them when to grow and proliferate, enables neurons to modulate reactions to neurotransmitters and allows liver cells to remove cholesterol from the bloodstream.
The system is so universally important in multicellular organisms that fungi, plants and animals all share virtually the same molecular machines to carry out endocytosis. Because of this uniformity, Hollopeter’s group works with the model organism C. elegans – a microscopic worm that is easy to manipulate – to figure out how the process works. They create different mutations that disrupt endocytosis, which causes distinctive fluid-filled cheeks that look like jowls. By figuring out how to fix the worms’ jowls and restore endocytosis, they can piece together which proteins are required for the system and what role they play. Then the researchers verify that the same rules apply in other animals using proteins isolated from humans and mice.
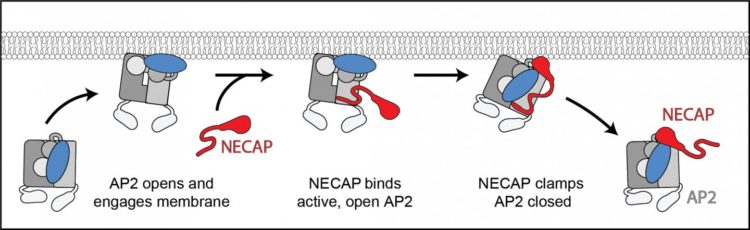
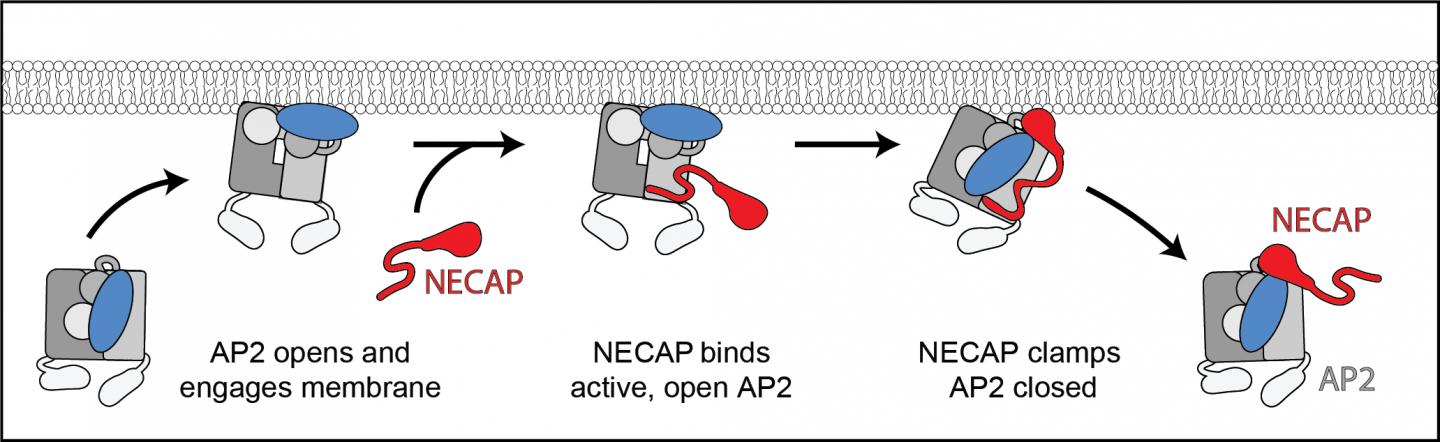
During endocytosis, cells form what looks like a “furry pit” under the macromolecules, engulf them and pinch off the payload within a membrane compartment inside the cell. The “fur” is actually a coat of scaffolding proteins called clathrin. Since clathrin can’t attach to the cell membrane directly, an adaptor protein complex called AP2 first changes into an open, active state and then links the scaffolding to the membrane to start the process.
Previously, Hollopeter’s group discovered that the protein FCHo acts like an accelerator on the process to open AP2 and that another protein, NECAP, acts as the brake and closes it. In their latest work, the team partnered with Dr. Richard Baker, a microscopist at the University of California, San Diego, to create a 3D visualization showing how NECAP interacts with AP2 using cryo-electron microscopy. This Nobel Prize-winning technology involves flash-freezing the proteins and using a high-energy electron beam to create an incredibly high-resolution image. The resulting visualizations show that NECAP works just like a chip clip on a bag of potato chips. It attaches on either side of the AP protein complex to clamp it shut and prevent endocytosis.
The researchers don’t know yet whether NECAP’s role is to help recycle the clathrin or if it turns off endocytosis as a form of quality control. “We think of it as acting either at the end of endocytosis,” said Partlow, “or at the beginning to turn it off, if the process starts at the wrong place or at the wrong time.”
The new findings directly contradict the work of other endocytosis researchers who also recently published a paper on NECAP’s function. They conclude that NECAP works as an accelerator instead of a brake. But Hollopeter and Partlow think that there are still components of endocytosis that the research community has yet to piece together.
In their current work, the group is investigating a new type of jowly worm, to figure out why disrupting endocytosis creates those chubby cheeks. Recently, Hollopeter and Baker received a five-year, $1.83 million grant from the National Institutes of Health to support their work to identify and visualize factors involved in endocytosis.
“Many people would say this has already been worked out and we know everything we need to know about it. But the more we look at endocytosis, the more we recognize that there’s still a lot we don’t know,” said Hollopeter. “We can use our system to get at fundamental questions of how this protein complex is regulated and that’s really an exciting opportunity.”
###
Media Contact
Melissa Osgood
[email protected]
607-255-9451
Original Source
https:/
Related Journal Article
http://dx.