Breast cancer remains a formidable adversary in women’s health worldwide, claiming countless lives annually despite advances in treatment. Central to effective management is the accurate identification and delineation of tumor boundaries within Dynamic Contrast-Enhanced Magnetic Resonance Imaging (DCE-MRI) scans. The intrinsic complexity of breast tumors, characterized by highly heterogeneous morphology, varying sizes, and diverse enhancement patterns over time, poses significant challenges for both radiologists and existing automated segmentation algorithms. Manual delineation not only demands extensive time and expertise but is also fraught with variability among observers, compromising consistency and reproducibility in clinical assessment.
In response to these challenges, a groundbreaking deep learning framework named Residual-Guided Spatiotemporal Transformer Graph Fusion (RST2G) has been developed to revolutionize breast tumor segmentation within DCE-MRI imaging. This innovative approach synergistically integrates residual learning mechanisms, hybrid feature extraction strategies, and the fusion of spatiotemporal graph representations. The design is meticulously tailored to leverage DCE-MRI’s multifaceted temporal and spatial data, combining pre-contrast and multiple post-contrast phases, enabling a more nuanced understanding of tumor vascular dynamics and tissue heterogeneity than conventional methods.
At the heart of RST2G lies the CFormerEncoder, a hybrid feature extractor that blends convolutional neural networks with transformer-based architectures. This amalgamation allows the model to capture both local fine-grained details and long-range dependencies across the volumetric and temporal dimensions of DCE-MRI data. The integration of residual-guided multi-scale refinement modules further enhances the model’s ability to resolve subtle and complex tumor boundaries by iteratively refining feature maps and preserving crucial anatomical detail. This refinement is bolstered by a unique spatiotemporal graph fusion component that models dynamic interactions between tissue regions over time, effectively synthesizing signal changes that signify tumor progression or response.
To validate their model, the researchers applied RST2G to two robust publicly available DCE-MRI datasets. The Breast-MRI-NACT-Pilot dataset comprises 64 patients undergoing neoadjuvant chemotherapy, encompassing multi-phase contrast scans that illuminate treatment-induced morphological changes. The TCGA-BRCA cohort, with 139 patients and extended post-contrast imaging, offered a substantial and diverse testbed representing various tumor phenotypes and contrast kinetics. Across these datasets, RST2G demonstrated striking performance gains, with Dice Similarity Coefficients reaching 61.8% and 80.1%, respectively, substantially surpassing traditional U-Net variants and 3D volumetric models.
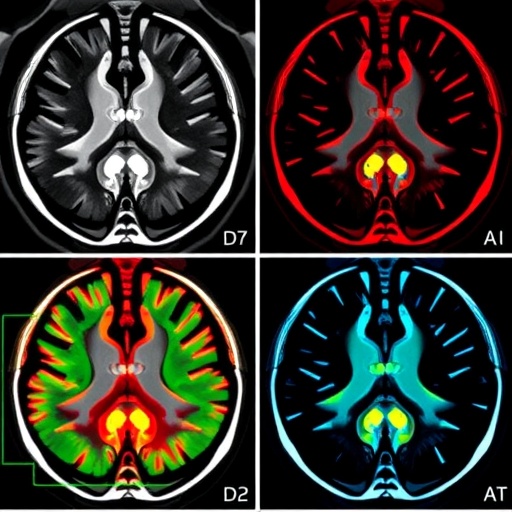
Beyond segmentation accuracy, the model excelled in minimizing relative volume difference (RVD), achieving near-ideal tumor volume quantification—critical for accurate treatment monitoring and prognosis assessment. Importantly, RST2G maintained strong generalization capabilities on external datasets acquired from clinical centers with distinct imaging protocols, underscoring its robustness and potential for real-world clinical adaptability. Visualizations produced using Grad-CAM techniques confirmed that the residual-guided attention mechanisms homed in on pathologically relevant tumor regions and boundaries, offering interpretability and fostering clinician confidence in the automated outputs.
The implications of RST2G’s success extend far beyond mere segmentation metrics. By automating tumor delineation in DCE-MRI, the framework alleviates the considerable workload burden on radiologists and reduces inter-observer variability, which has historically plagued precision oncology workflows. Accurate and consistent tumor boundary definition facilitates precise volumetric measurement, essential for planning surgical interventions and evaluating neoadjuvant chemotherapy efficacy. Moreover, the ability to capture spatiotemporal tumor dynamics opens new avenues for personalized treatment strategies and longitudinal disease monitoring.
Technically, RST2G is designed with clinical translation in mind. The entire processing pipeline can segment a full MRI volume in approximately 30 seconds using a standard 10 GB GPU, making near-real-time deployment feasible in busy radiology departments. This computational efficiency stems from the model’s hybrid transformer-graph architecture, which harnesses the parallelization strengths of attention mechanisms while mitigating computational overhead via graph fusion techniques. Such efficiency is pivotal in integrating AI-assisted tools seamlessly into established clinical imaging workflows.
Looking ahead, the research team aims to rigorously validate RST2G across multi-center cohorts to ensure robustness against heterogeneous imaging protocols, scanner types, and patient populations—a critical step for regulatory approval and widespread adoption. Another exciting frontier lies in adapting the model to irregular temporal sampling inherent in DCE-MRI acquisitions, enabling flexible analysis regardless of varying post-contrast scan timings. Integration with clinical decision-support systems is also planned to deliver actionable information directly to oncologists and multidisciplinary teams, thus advancing personalized breast cancer care.
In essence, RST2G represents a paradigm shift in automated breast tumor segmentation by explicitly modeling the complex spatiotemporal trajectories of contrast agent dynamics in DCE-MRI. Its novel fusion of residual learning, hybrid feature extraction, and graph-based temporal modeling surmounts previous limitations encountered by conventional networks. As a result, the framework offers unprecedented accuracy, interpretability, and clinical readiness, promising to transform how breast cancer imaging data is analyzed and leveraged for therapeutic decision-making.
The authors of this innovative study assert that by enhancing segmentation precision and consistency, RST2G addresses an urgent unmet need, facilitating more reliable assessments of tumor morphology and treatment response in clinical practice. This breakthrough technique not only holds promise for improving diagnostic workflows but also serves as a foundational platform upon which future AI-driven imaging innovations can be built.
—
Subject of Research: Breast cancer tumor segmentation in Dynamic Contrast-Enhanced MRI using deep learning
Article Title: RST2G: Residual-Guided Spatiotemporal Transformer Graph Fusion Enhancement for Breast Cancer Segmentation in DCE-MRI
News Publication Date: March 23, 2026
Web References: https://doi.org/10.34133/cbsystems.0502
Image Credits: Maoshan Chen, Department of Breast and Thyroid Surgery, Suining Central Hospital
Keywords
Breast Cancer, Tumor Segmentation, DCE-MRI, Deep Learning, Spatiotemporal Modeling, Residual Learning, Transformer Networks, Graph Fusion, Medical Imaging, AI in Healthcare, Oncology, Neural Networks
Tags: automated breast tumor segmentationbreast cancer imaging advancementsbreast tumor vascular dynamics analysisclinical applications of RST2Gconvolutional-transformer neural networksDCE-MRI tumor segmentationdeep learning in medical imaginghybrid feature extraction for MRIimproving MRI segmentation accuracyresidual-guided spatiotemporal transformerspatiotemporal graph fusion modelstumor boundary delineation techniques