A recent study explains an essential component for proper mitochondrial function: The protein complexes MICOS and ATP synthase can communicate with each other. Dr. Heike Rampelt and Prof. Dr. Nikolaus Pfanner at the Institute of Biochemistry and Molecular Biology of the University of Freiburg have uncovered an important mechanism that ensures efficient metabolic adaptation of mitochondria. The research is a collaboration with the groups of Prof. Dr. Martin van der Laan of Saarland University, Prof. Dr. Claudine Kraft of the University of Freiburg and Prof. Dr. Ida van der Klei of the University Groningen / Netherlands and combines biochemical approaches with fluorescence microscopy of living cells as well as electron microscopy to visualize mitochondrial membrane architecture. The study has been published in the journal Cell Reports.
Cellular respiration of the inner mitochondrial membrane
Mitochondria, the power plants of the cell, make massive contributions to the energy supply of the body by burning metabolites with the help of oxygen. This cellular respiration takes place in the inner of the two mitochondrial membranes that, in contrast to the outer membrane, is strongly folded. The structure and topology of these membrane folds, the so-called cristae membranes, has profound influence on the efficiency of respiration and is important for many mitochondrial functions. For this reason, cristae architecture is controlled precisely and adapted dynamically to changes in cellular metabolism. Defects in these processes result in severe human diseases.
Communication is key
Two protein complexes in the inner mitochondrial membrane that are required for a normal membrane architecture are the F1Fo-ATP synthase, an enzyme that also participates in energy conversion, and the MICOS complex (mitochondrial contact site and cristae organizing system). These complexes are regarded as antagonists; they are localized in different areas of the inner membrane and bend the membrane in opposite directions. It was unclear how the functions of these two protein complexes can be coordinated with each other. The team around Rampelt and Pfanner now demonstrate that MICOS and ATP synthase communicate with each other and that this is vital for healthy mitochondrial function. A MICOS subunit, Mic10, travels to the ATP synthase and stabilizes the association of several ATP synthases to large complexes. This new regulatory function of Mic10 is pivotal for efficient metabolic adaptation and respiratory growth. “Communication between the two complexes is likely key to the coordinated biogenesis of the inner mitochondrial membrane”, explains Rampelt.
Heike Rampelt, Nikolaus Pfanner and Claudine Kraft lead research groups at the Institute of Biochemistry and Molecular Biology of the Medical Faculty and perform research in the Excellence Cluster CIBSS of the University Freiburg in the area of biological signalling studies.
Original publication:
Rampelt, H., Wollweber, F., Licheva, M., de Boer, R., Perschil, I., Steidle, L., Becker, T., Bohnert, M., van der Klei, I., Kraft, C., van der Laan, M., Pfanner, N. (2022): Dual role of Mic10 in mitochondrial cristae organization and ATP synthase-linked metabolic adaptation and respiratory growth. In: Cell Reports, 38:110290. DOI: https://doi.org/10.1016/j.celrep.2021.110290
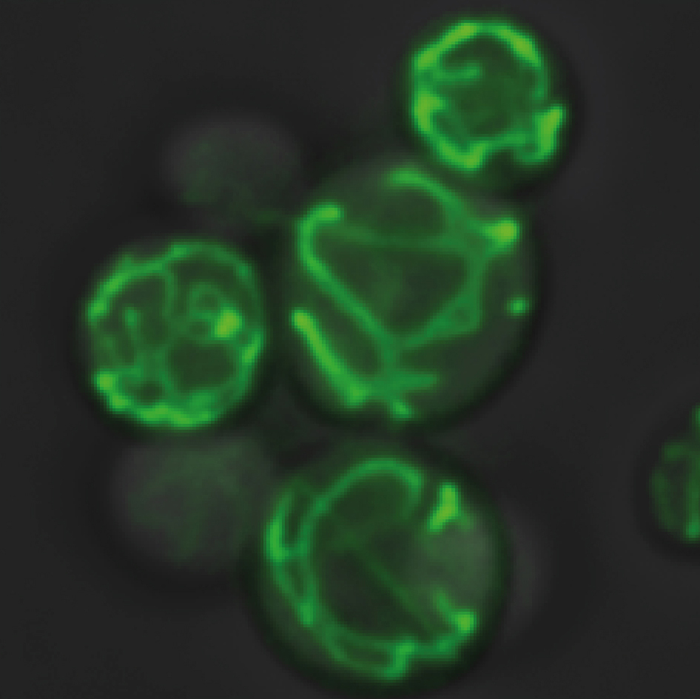
Credit: Mariya Licheva, University of Freiburg
A recent study explains an essential component for proper mitochondrial function: The protein complexes MICOS and ATP synthase can communicate with each other. Dr. Heike Rampelt and Prof. Dr. Nikolaus Pfanner at the Institute of Biochemistry and Molecular Biology of the University of Freiburg have uncovered an important mechanism that ensures efficient metabolic adaptation of mitochondria. The research is a collaboration with the groups of Prof. Dr. Martin van der Laan of Saarland University, Prof. Dr. Claudine Kraft of the University of Freiburg and Prof. Dr. Ida van der Klei of the University Groningen / Netherlands and combines biochemical approaches with fluorescence microscopy of living cells as well as electron microscopy to visualize mitochondrial membrane architecture. The study has been published in the journal Cell Reports.
Cellular respiration of the inner mitochondrial membrane
Mitochondria, the power plants of the cell, make massive contributions to the energy supply of the body by burning metabolites with the help of oxygen. This cellular respiration takes place in the inner of the two mitochondrial membranes that, in contrast to the outer membrane, is strongly folded. The structure and topology of these membrane folds, the so-called cristae membranes, has profound influence on the efficiency of respiration and is important for many mitochondrial functions. For this reason, cristae architecture is controlled precisely and adapted dynamically to changes in cellular metabolism. Defects in these processes result in severe human diseases.
Communication is key
Two protein complexes in the inner mitochondrial membrane that are required for a normal membrane architecture are the F1Fo-ATP synthase, an enzyme that also participates in energy conversion, and the MICOS complex (mitochondrial contact site and cristae organizing system). These complexes are regarded as antagonists; they are localized in different areas of the inner membrane and bend the membrane in opposite directions. It was unclear how the functions of these two protein complexes can be coordinated with each other. The team around Rampelt and Pfanner now demonstrate that MICOS and ATP synthase communicate with each other and that this is vital for healthy mitochondrial function. A MICOS subunit, Mic10, travels to the ATP synthase and stabilizes the association of several ATP synthases to large complexes. This new regulatory function of Mic10 is pivotal for efficient metabolic adaptation and respiratory growth. “Communication between the two complexes is likely key to the coordinated biogenesis of the inner mitochondrial membrane”, explains Rampelt.
Heike Rampelt, Nikolaus Pfanner and Claudine Kraft lead research groups at the Institute of Biochemistry and Molecular Biology of the Medical Faculty and perform research in the Excellence Cluster CIBSS of the University Freiburg in the area of biological signalling studies.
Original publication:
Rampelt, H., Wollweber, F., Licheva, M., de Boer, R., Perschil, I., Steidle, L., Becker, T., Bohnert, M., van der Klei, I., Kraft, C., van der Laan, M., Pfanner, N. (2022): Dual role of Mic10 in mitochondrial cristae organization and ATP synthase-linked metabolic adaptation and respiratory growth. In: Cell Reports, 38:110290. DOI: https://doi.org/10.1016/j.celrep.2021.110290
Contact:
Dr. Heike Rampelt
Institute of Biochemistry and Molecular Biology
University of Freiburg
Phone: +49 (0)761 / 203 – 5245
E-Mail: [email protected]
Annette Kollefrath-Persch
Office of University and Science Communications
University of Freiburg
Phone: +49 (0)761 / 203 – 8909
E-Mail: [email protected]
DOI
10.1016/j.celrep.2021.110290